Understanding Your Glucose Metrics for Better Diabetes Control In 2026
Learn to interpret glucose metrics like TIR, HbA1c, and GMI. Understand normal ranges and variability for better control in 2026.
Written by Dr. Rishav Das, M.B.B.S. — Wellness Device Data Analyst | Health Informatics Specialist
Medically reviewed according to the standards outlined on our editorial standards page
❌ Confused by your CGM report? ❌ Don’t know if your levels are actually “good” for you specifically? ❌ Overwhelmed by HbA1c, Time in Range, and TAR?
You’re not alone. Most people with diabetes feel lost when looking at their glucose data. This guide translates HbA1c, Time in Range, and CGM metrics into actionable insights—so you can prevent complications, reduce medication adjustments, and feel confident discussing results with your doctor.
Why understanding these metrics matters:
This isn’t just about numbers—it’s about preventing vision loss, protecting your kidneys, and enjoying life without constant worry about blood sugar emergencies. Research shows each 5% improvement in Time in Range reduces your risk of diabetic retinopathy by 64%. Better glucose control means fewer unexpected lows, more predictable energy throughout the day, and reduced anxiety about what’s happening inside your body.
⏱️ Need Quick Answers? Jump to:
Normal Blood Sugar Ranges | What’s a Good HbA1c? | Time in Range Targets | When to Call Your Doctor | Do You Need a CGM?
Blood glucose monitoring produces multiple metrics that provide different insights into glycemic control. This page explains the most clinically significant measurements and how to interpret them in consultation with your healthcare provider.
- Understanding Your Glucose Metrics for Better Diabetes Control In 2026
- Blood Glucose Basics
- Do You Need a CGM? Cost-Benefit Analysis
- Time in Range (TIR)
- Glucose Variability
- HbA1c (Glycated Hemoglobin)
- CGM-Specific Metrics
- Reading Your Glucose Report
- When to Contact Your Doctor
- How to Discuss These Metrics with Your Doctor
- Frequently Asked Questions
- What is a good Time in Range for Type 2 diabetes?
- Is 7.5% HbA1c bad?
- How much does a CGM cost without insurance?
- Can I improve Time in Range without medication changes?
- What's the difference between HbA1c and GMI?
- How do I know if my glucose monitor is accurate?
- What does it mean if my HbA1c is 7.0% but my GMI is 7.8%?
- Should I check my blood sugar before or after exercise?
- What's considered dangerously high blood sugar?
- How can I reduce my HbA1c naturally?
- Does the time of day I check my blood sugar matter?
- What should my blood sugar be at bedtime?
- How often should I check my HbA1c?
- Can stress affect my blood sugar?
- What's the best app for tracking Time in Range?
- How do I explain my glucose data to my doctor in 5 minutes or less?
- References
Blood Glucose Basics
Normal Blood Sugar Ranges
Blood glucose levels are measured in milligrams per deciliter (mg/dL) in the United States. Target ranges vary based on timing, diabetes status, and individual health factors.
| Population | Fasting (mg/dL) | 2-Hour Postprandial (mg/dL) | Random (mg/dL) |
| Non-diabetic | 70-99 | <140 | <200 |
| Prediabetes | 100-125 | 140-199 | — |
| Diabetes (general) | 80-130 | <180 | Varies |
| Diabetes (individualized) | Per provider guidance | Per provider guidance | Per provider guidance |
Source: American Diabetes Association (ADA) Standards of Medical Care in Diabetes, 2024
Normal Blood Sugar Ranges by Age and Diabetes Status
Blood sugar targets can vary based on age and individual health factors. While the general diabetes target is 80-130 mg/dL fasting and <180 mg/dL post-meal, these ranges may be adjusted:
Adults Under 40 with Diabetes:
- Often have tighter targets (80-130 fasting, <140 post-meal) if achievable without hypoglycemia
- Longer life expectancy means greater benefit from strict control
- Lower threshold for starting medication intensification
Adults 40-65 with Diabetes:
- Standard targets apply (80-130 fasting, <180 post-meal)
- Individualized based on comorbidities and hypoglycemia history
- Balance between complication prevention and quality of life
Adults Over 65 with Diabetes:
- May have less stringent targets (90-150 fasting, <200 post-meal) if:
- Limited life expectancy (<10 years)
- History of severe hypoglycemia
- Multiple comorbidities or frailty
- Prioritize avoiding hypoglycemia over tight control
- Discuss individualized targets with your provider
Children and Adolescents with Diabetes:
- Targets often slightly higher to avoid hypoglycemia during growth periods
- Consult pediatric endocrinologist for age-appropriate ranges
- Balance between glycemic control and normal development
Pregnant Individuals:
- Tightest targets of all groups (fasting <95 mg/dL, 1-hour post-meal <140 mg/dL, 2-hour <120 mg/dL)
- Protect fetal development and reduce obstetric complications
- Require specialized prenatal care and frequent monitoring
Source: ADA Standards of Care 2024; AGS Diabetes Guidelines for Older Adults
Key considerations:
- Pregnant individuals require tighter glycemic control with specialized targets
- Individual targets may differ based on age, comorbidities, hypoglycemia risk, and diabetes duration
- Older adults and those with cardiovascular disease may have less stringent targets
Fasting vs Post-Meal Glucose
Glucose measurements capture different physiological states depending on timing relative to food intake.
| Measurement Type | Definition | Clinical Significance | Typical Testing Window |
| Fasting Plasma Glucose (FPG) | Glucose level after 8+ hours without caloric intake | Reflects basal glucose production by the liver; used for diagnosis | Morning, before breakfast |
| Postprandial Glucose (PPG) | Glucose level after eating | Reflects insulin response to carbohydrate intake; associated with cardiovascular risk | 1-2 hours after meal start |
| Random Glucose | Glucose measured at any time | Used for screening or symptom evaluation | No timing requirement |
| Preprandial Glucose | Glucose before meals | Guides mealtime insulin dosing | Immediately before eating |
Sources: ADA Standards of Care; International Diabetes Federation (IDF) Postprandial Glucose Guidelines
Why both matter:
- Postprandial hyperglycemia is independently associated with cardiovascular disease risk, even when fasting levels are controlled
- Elevated fasting glucose may indicate insufficient overnight insulin production or excessive hepatic glucose output
- Elevated postprandial glucose may suggest inadequate mealtime insulin response
Factors That Affect Blood Sugar

Multiple physiological and behavioral factors influence glucose levels beyond food intake.
Dietary Factors:
- Carbohydrate quantity and type (simple vs complex, glycemic index)
- Macronutrient composition (protein and fat slow glucose absorption)
- Fiber content (soluble fiber delays gastric emptying)
- Meal timing and frequency
- Alcohol consumption (may cause delayed hypoglycemia)
Physical Activity:
- Aerobic exercise typically lowers glucose during and after activity
- Resistance training may initially raise glucose, then lower it
- High-intensity interval training (HIIT) can cause transient glucose elevation
- Exercise timing relative to meals affects response magnitude
Medications:
- Diabetes medications (insulin, metformin, sulfonylureas, GLP-1 agonists, SGLT2 inhibitors)
- Corticosteroids (prednisone, dexamethasone) elevate glucose
- Beta-blockers may mask hypoglycemia symptoms
- Certain antipsychotics and immunosuppressants affect glucose metabolism
Physiological Factors:
- Somogyi effect (rebound hyperglycemia after nocturnal hypoglycemia)
- Stress hormones (cortisol, epinephrine) raise glucose
- Illness and infection trigger counter-regulatory responses
- Menstrual cycle hormones affect insulin sensitivity
- Sleep duration and quality (poor sleep associated with insulin resistance)
- Dawn phenomenon (pre-dawn cortisol surge raises morning glucose)
Do You Need a CGM? Cost-Benefit Analysis
Continuous glucose monitors (CGMs) typically cost $150-$300 per month without insurance coverage. That’s a significant expense—so is it worth it?
CGM vs. Traditional Fingerstick Monitoring:
| Aspect | Traditional Meter | Continuous Glucose Monitor (CGM) |
|---|---|---|
| Cost | $30-50/month (test strips) | $150-300/month (varies by device and insurance) |
| Measurements | 4-10 per day (manual) | 288+ per day (automatic every 5 minutes) |
| Trend Detection | Limited | Real-time trends and alerts |
| Hypoglycemia Alerts | None | Predictive alerts 15-30 minutes before lows |
| Time in Range | Cannot calculate | Automatically tracked |
| Insurance Coverage | Most plans cover | Covered for Type 1; Type 2 coverage expanding |
Who benefits most from CGM:
- Type 1 diabetes (any age)
- Type 2 diabetes on intensive insulin therapy
- History of severe hypoglycemia or hypoglycemia unawareness
- HbA1c above target despite medication adherence
- High glucose variability (frequent swings between highs and lows)
- Pregnancy with diabetes (any type)
- Difficulty achieving Time in Range goals with fingersticks alone
These recommendations align with clinical guidelines for CGM candidacy published in Diabetes Care, which evaluated evidence across multiple patient populations.
Patient Perspective:
“I resisted getting a CGM for 2 years because of the $200/month cost. Within the first month, it caught a pattern of overnight lows I never knew I had—I was waking up at 250 mg/dL every morning because my body was rebounding from 3 AM lows. That one insight eliminated 6 months of frustration trying to figure out my morning highs. The cost pays for itself in avoiding complications and ER visits.”
— James T., Type 2 diabetes, 14 years
Cost vs. complication savings:
Research shows CGM users reduce their annual emergency room visits by an average of 1-2 visits (saving $2,000-$5,000), decrease severe hypoglycemia events by 40-60%, and may reduce long-term complication costs by up to $9,000 annually through better glycemic control. For many, the monthly CGM cost is offset within the first year through reduced medical emergencies and complications.
Insurance coverage tips:
- Medicare covers CGM for insulin-dependent diabetes (Part B)
- Most private insurance covers CGM for Type 1 diabetes
- Type 2 diabetes coverage requires documentation of intensive insulin therapy (3+ injections daily) or hypoglycemia history
- Prior authorization usually requires 3-6 months of blood glucose logs showing inadequate control
- Appeal denials with your doctor’s letter citing clinical necessity
Not ready for CGM?
You can still estimate Time in Range with 7-10 fingerstick measurements daily using this approach: Check fasting, pre-meals, 2-hours post-meals, and bedtime. Calculate what percentage fall within 70-180 mg/dL. While less accurate than CGM, this provides a baseline TIR estimate to discuss with your provider.
Top CGM Devices Compared (2026):
| Device | Dexcom G7 | Freestyle Libre 3 | Medtronic Guardian 4 | Eversense E3 |
|---|---|---|---|---|
| Wear Time | 10 days | 14 days | 7 days | 6 months (implanted) |
| Cost (no insurance) | $240-300/month | $150-200/month | $250-300/month | $300/month + procedure |
| Accuracy (MARD) | 8.2% | 7.9% | 8.5% | 8.5% |
| Phone Compatibility | iOS & Android | iOS & Android | iOS & Android | iOS & Android |
| Alerts | Predictive (20 min) | Real-time | Predictive (30 min) | Real-time |
| Best For | Most users | Budget-conscious; no calibration | Pump integration | Avoids frequent sensors |
| Medicare Coverage | Yes | Yes | Yes | No (2026) |
Accuracy measured as Mean Absolute Relative Difference (MARD). Lower = better. All devices FDA-approved for treatment decisions without confirmatory fingersticks.
Which CGM is right for you?
Take this 60-second assessment:
- Do you use an insulin pump? → Guardian 4 (seamless integration) or Dexcom G7 (works with most pumps)
- Budget is your main concern? → Freestyle Libre 3 (lowest cost, no calibration)
- Want fewest sensor changes? → Eversense E3 (6 months, requires minor procedure)
- Need the most accurate readings? → Freestyle Libre 3 (7.9% MARD, best current accuracy)
Next step: Discuss CGM options with your endocrinologist or diabetes educator. Bring this comparison table to your appointment.
Time in Range (TIR)
What Time in Range Measures
Time in Range (TIR) represents the percentage of time glucose levels remain within a target range, typically 70-180 mg/dL for most adults with diabetes.
| Metric | Definition | Standard Target Range | Measurement Period |
| Time in Range (TIR) | % of glucose readings between 70-180 mg/dL | 70-180 mg/dL (general) | Typically 14 days minimum |
| Time Below Range (TBR) | % of readings <70 mg/dL | <70 mg/dL (Level 1 hypoglycemia) | Continuous monitoring period |
| Time in Tight Range | % of readings between 70-140 mg/dL | 70-140 mg/dL (pregnancy, some Type 1) | Specialized populations |
| Time Above Range (TAR) | % of readings >180 mg/dL | >180 mg/dL (Level 1 hyperglycemia) | Continuous monitoring period |
Source: International Consensus on Time in Range (Battelino et al., Diabetes Care, 2019) This 70-180 mg/dL range and the >70% target are based on guidelines published in Diabetes Care by Battelino and colleagues, which analyzed data from thousands of CGM users to establish evidence-based thresholds
Why TIR emerged:
- TIR correlates with microvascular complications risk (retinopathy, nephropathy, neuropathy)
- Continuous glucose monitors (CGMs) provide thousands of data points beyond single daily measurements
- Average glucose alone does not capture glycemic variability
All device accuracy claims on this site are evaluated using our published testing methodology.
Real Example: How TIR Changed Treatment
“After I started using a CGM, I learned my Time in Range was only 52%—I thought I was doing fine because my HbA1c was 7.4%. My doctor and I adjusted my basal insulin and meal timing. Three months later, my TIR is 78% and my HbA1c dropped to 6.9%. More importantly, I stopped having afternoon crashes and now I can predict how I’ll feel each day.”
— Maria L., Type 1 diabetes, 8 years
Target TIR Percentages
Clinical consensus recommends specific TIR goals based on diabetes type and individual risk factors.
| Population | TIR Goal (70-180 mg/dL) | TBR <70 mg/dL | TBR <54 mg/dL | Rationale |
| Type 1 Diabetes | >70% | <4% | <1% | Balances glycemic control with hypoglycemia risk |
| Type 2 Diabetes | >70% | <4% | <1% | Same targets as Type 1 per consensus |
| Pregnancy (all types) | >70% (63-140 mg/dL range) | <4% | <1% | Tighter targets protect fetal development |
| Older/High-Risk Adults | >50% | <1% | <0.5% | Prioritizes hypoglycemia avoidance |
Source: Battelino T, et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation. Diabetes Care. 2019;42(8):1593-1603.
Clinical interpretation:
- High TBR (>4%) indicates dangerous hypoglycemia patterns requiring immediate intervention
- Each 5% increase in TIR is associated with reduced complication risk
- Less than 70% TIR suggests need for therapy adjustment
Why TIR Matters More Than Averages
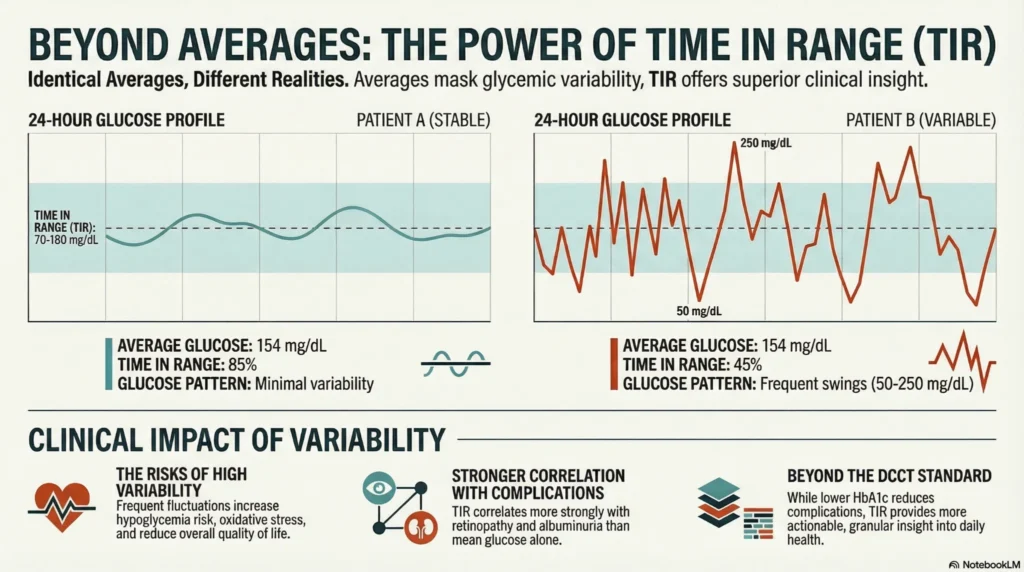
Average glucose can be identical across different glycemic patterns, but clinical outcomes differ substantially.
Example Scenario:
| Patient | Average Glucose (mg/dL) | TIR (70-180 mg/dL) | Pattern Description |
| Patient A | 154 | 85% | Stable glucose with minimal variability |
| Patient B | 154 | 45% | Frequent swings between 50 and 250 mg/dL |
Both have the same average, but Patient B experiences:
- Higher hypoglycemia risk (symptoms, impaired cognition, cardiovascular events)
- Greater oxidative stress from variability
- Reduced quality of life from glucose fluctuations
- Potentially higher long-term complication risk
Evidence base:
- The DCCT (Diabetes Control and Complications Trial) demonstrated that lower HbA1c reduces complications, but TIR provides more granular insight
- Studies show TIR correlates more strongly with retinopathy and albuminuria than mean glucose alone
- Glucose variability independently predicts cardiovascular events in some populations
Sources: DCCT Research Group, N Engl J Med, 1993; Beck RW, et al., Diabetes Care, 2019
💬 Share This:
“Your average glucose can be perfect, but if you’re swinging between 50 and 250 mg/dL daily, you’re still at high risk for complications. Time in Range (TIR) captures what averages miss—glucose stability.” #DiabetesAwareness #CGM
💡 Key Takeaway: Why Time in Range Matters
Each 5% increase in Time in Range is associated with a 64% lower risk of diabetic retinopathy. Better TIR means:
- Fewer unexpected low blood sugar episodes
- More predictable energy throughout the day
- Less anxiety about “what’s happening” between meter checks
- Reduced long-term risk of vision loss, kidney disease, and nerve damage
Bottom line: Two people with the same HbA1c can have vastly different complication risks based on their glucose stability.
Glucose Variability
Standard Deviation
Standard deviation (SD) quantifies the spread of glucose values around the mean, measured in mg/dL.
| SD Range (mg/dL) | Interpretation | Clinical Implication |
| <50 | Low variability | Stable glucose control; reduced hypoglycemia risk |
| 50-75 | Moderate variability | Acceptable for many individuals; monitor trends |
| >75 | High variability | Consider therapy adjustment; assess hypoglycemia patterns |
Calculation context:
- SD represents how much individual readings deviate from the average
- Higher SD indicates more glucose fluctuations
- SD should be interpreted alongside mean glucose (a 150 mg/dL average with 80 mg/dL SD differs from 200 mg/dL average with 30 mg/dL SD)
Limitations:
- Less intuitive than percentage-based metrics for patients
- Does not distinguish between hypoglycemic vs. hyperglycemic excursions
- Sensitive to extreme outliers
Coefficient of Variation
Coefficient of Variation (CV) expresses variability as a percentage of the mean, providing context-adjusted measurement.
| Metric | Formula | Target | Interpretation |
| CV (%) | (Standard Deviation ÷ Mean Glucose) × 100 | ≤36% | Standardized variability metric |
| Stable glucose | CV <36% | Recommended | Low variability relative to mean |
| Unstable glucose | CV >36% | Action needed | High variability; therapy review indicated |
Source: Danne T, et al. International Consensus on Use of CGM. Diabetes Care. 2017;40(12):1631-1640.
Advantages over SD:
- Allows comparison across different mean glucose levels
- 36% threshold is consistent regardless of average glucose
- More clinically interpretable for provider-patient discussions
Example:
- Patient A: Mean 120 mg/dL, SD 50 mg/dL → CV = 42% (high variability)
- Patient B: Mean 180 mg/dL, SD 50 mg/dL → CV = 28% (acceptable variability)
Both have the same SD, but Patient A has problematic variability relative to their tighter control.
Reducing Blood Sugar Spikes
Strategies to minimize postprandial glucose excursions and overall variability.
Dietary Interventions:
- Carbohydrate distribution — Spread carbohydrate intake across meals rather than concentrating in single servings
- Low glycemic index foods — Choose whole grains, legumes, and non-starchy vegetables over refined carbohydrates
- Macronutrient pairing — Combine carbohydrates with protein and healthy fats to slow absorption
- Vinegar consumption — Evidence suggests 1-2 tablespoons before meals may reduce postprandial spikes
- Meal sequence — Some studies indicate eating vegetables/protein before carbohydrates reduces glucose elevation
Physical Activity Timing:
- Post-meal walking — 10-15 minutes of light activity after eating may reduce glucose peaks
- Resistance exercise — Improves insulin sensitivity over time
- Timing insulin boluses — Pre-bolusing 15-20 minutes before meals (Type 1 diabetes, under medical supervision)
Medication Optimization:
- Rapid-acting insulin analogs — Match insulin peak to food absorption
- GLP-1 receptor agonists — Slow gastric emptying and reduce postprandial glucose
- Alpha-glucosidase inhibitors — Delay carbohydrate digestion
Technology-Assisted Strategies:
- Continuous glucose monitors (CGM) — Real-time feedback enables behavioral adjustments
- Automated insulin delivery systems — Algorithm-driven insulin dosing reduces variability
- Carbohydrate counting apps — Improve bolus accuracy
Sources: American Diabetes Association; Shukla AP, et al., Diabetologia, 2017; Colberg SR, et al., Diabetes Care, 2016
⚠️ Clinical Note: All medication and insulin adjustments should be made under healthcare provider supervision.
HbA1c (Glycated Hemoglobin)
What HbA1c Measures
HbA1c (glycated hemoglobin, also written as A1C or hemoglobin A1c) reflects average blood glucose over approximately 2-3 months.
| Aspect | Description |
| Biochemical process | Glucose molecules attach irreversibly to hemoglobin in red blood cells through non-enzymatic glycation |
| Measurement period | Reflects average glucose over 2-3 months (lifespan of red blood cells) |
| Units | Reported as percentage (%) or mmol/mol (IFCC standard) |
| Testing frequency | Every 3-6 months for individuals with diabetes; annually for prediabetes screening |
| Sample type | Venous blood draw or fingerstick (point-of-care devices) |
Diagnostic criteria:
| HbA1c Level | Classification | Interpretation |
| <5.7% | Normal | No diabetes |
| 5.7-6.4% | Prediabetes | Increased diabetes risk; lifestyle intervention recommended |
| ≥6.5% | Diabetes | Diagnostic threshold (requires confirmation) |
Source: American Diabetes Association Standards of Medical Care in Diabetes, 2024
Advantages:
- No fasting required
- Reflects long-term control rather than daily fluctuations
- Standardized internationally
- Strong correlation with complications risk in clinical trials
Limitations:
- Lag time means recent changes not immediately visible
- Does not capture glucose variability or hypoglycemia
- Affected by conditions altering red blood cell lifespan (anemia, hemoglobinopathies, kidney disease)
- May not accurately reflect control in certain ethnic populations
HbA1c Targets by Diabetes Type
Individualized targets balance glycemic control benefits against hypoglycemia risk and treatment burden.
| Population | General Target | Considerations | Less Stringent Target |
| Type 1 Diabetes (adults) | <7.0% | Minimize hypoglycemia; CGM use helpful | <7.5-8.0% if severe hypoglycemia history |
| Type 2 Diabetes (general) | <7.0% | Most adults without complicating factors | 7.5-8.0% for older adults, limited life expectancy |
| Type 2 Diabetes (newly diagnosed) | <6.5% | May prevent progression; if achievable without hypoglycemia | — |
| Pregnancy (preexisting diabetes) | <6.0% (ideally) | Protect fetal development; tight monitoring | Individualized if hypoglycemia risk high |
| Gestational diabetes | <6.0% | Reduce obstetric complications | — |
| Children/Adolescents (Type 1) | <7.0% | Balance control with developmental needs | <7.5% if recurrent severe hypoglycemia |
Source: ADA Standards of Care in Diabetes, 2024; NICE Diabetes Guidelines per ADA’s latest evidence-based recommendations
Factors favoring less stringent targets:
- Limited life expectancy (<10 years)
- Advanced complications (severe cardiovascular disease, advanced kidney disease)
- Extensive comorbidities
- Recurrent severe hypoglycemia or hypoglycemia unawareness
- Individual patient preferences and treatment burden concerns
Factors favoring more stringent targets (if achievable safely):
- Patient motivation and resources for intensive management
- Shorter diabetes duration
- Younger age with longer life expectancy
- No significant cardiovascular disease
- Absence of hypoglycemia risk
Common Question: “My A1c is 7.2%—is that good or bad?”
The answer depends on YOUR individualized target. A 7.2% HbA1c could be:
- Above goal if your target is <7.0% (indicating need for adjustment)
- At goal if you’re older with other health conditions (7.0-7.5% range)
- Excellent if you’ve been running 9-10% and recently lowered it
Ask your doctor: “What is MY specific HbA1c target, and why?” Targets vary based on age, how long you’ve had diabetes, other health conditions, and hypoglycemia risk. There’s no universal “good” number—only what’s appropriate for your situation.
Relationship to Average Glucose
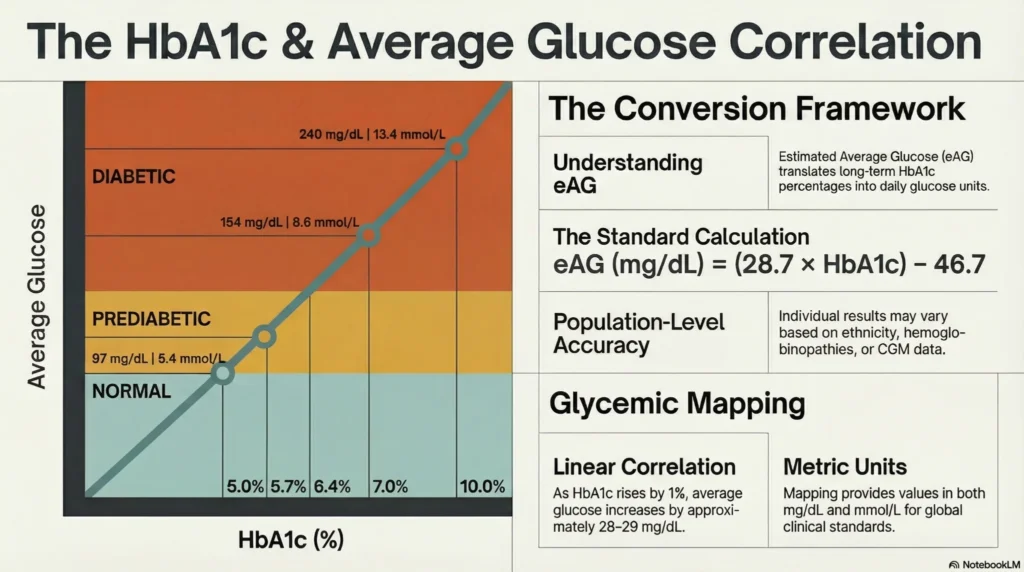
HbA1c correlates with average glucose, allowing estimation of mean glucose from HbA1c values.
HbA1c to Estimated Average Glucose (eAG) Conversion:
| HbA1c (%) | Estimated Average Glucose (mg/dL) | Estimated Average Glucose (mmol/L) |
| 5.0% | 97 | 5.4 |
| 5.5% | 111 | 6.2 |
| 6.0% | 126 | 7.0 |
| 6.5% | 140 | 7.8 |
| 7.0% | 154 | 8.6 |
| 7.5% | 169 | 9.4 |
| 8.0% | 183 | 10.2 |
| 8.5% | 197 | 11.0 |
| 9.0% | 212 | 11.8 |
| 9.5% | 226 | 12.6 |
| 10.0% | 240 | 13.4 |
Formula: eAG (mg/dL) = (28.7 × HbA1c) − 46.7
Source: Nathan DM, et al. Translating the A1C Assay Into Estimated Average Glucose Values. Diabetes Care. 2008;31(8):1473-1478. The conversion between HbA1c and estimated average glucose is calculated using research translating the A1C assay into average glucose values, which established the mathematical relationship through analysis of continuous glucose data
Important caveats:
- Recent CGM data (Glucose Management Indicator) may provide more accurate individual estimates
- This is a population-level correlation; individual variation exists
- HbA1c may overestimate average glucose in some populations (e.g., certain ethnic groups, hemoglobinopathies)
📊 Pin This: Blood Sugar & HbA1c Conversion Chart
[This table is optimized for Pinterest and Instagram sharing. Users can screenshot or download for quick reference.]Save this chart to your phone or print it for your fridge—it’s the fastest way to understand what your HbA1c means in daily blood sugar terms.
CGM-Specific Metrics
Glucose Management Indicator (GMI)
GMI estimates what HbA1c would be based on CGM average glucose over 14 days, using the same mathematical relationship as eAG.
| Metric | Formula | Interpretation | Use Case |
| GMI (%) | (3.31 + 0.02392 × mean CGM glucose) | Estimated HbA1c from CGM data | Compare CGM-derived control to lab HbA1c |
Source: Bergenstal RM, et al. Diabetes Care. 2018;41(11):2275-2280.
Key distinctions from HbA1c:
- GMI uses 14-day CGM average; HbA1c reflects 2-3 months
- GMI unaffected by red blood cell disorders
- Discrepancies between GMI and lab HbA1c may indicate:
- Recent control changes
- Biological factors affecting HbA1c measurement
- Insufficient CGM wear time (<70% of 14 days)
Clinical application:
- GMI provides near-real-time feedback between quarterly HbA1c tests
- Expect GMI within ±0.5% of lab HbA1c in most individuals
- Larger differences warrant investigation (recent behavior changes, hemoglobinopathy, kidney disease)
Time Below Range

Time Below Range (TBR) quantifies hypoglycemia exposure, a critical safety metric.
Hypoglycemia Categories:
| Level | Glucose Threshold | Clinical Significance | Target TBR |
| Level 1 (Alert) | <70 mg/dL (3.9 mmol/L) | Physiological threshold; autonomic symptoms may begin | <4% (58 minutes/day) |
| Level 2 (Serious) | <54 mg/dL (3.0 mmol/L) | Clinically significant; neuroglycopenic symptoms likely | <1% (15 minutes/day) |
| Level 3 (Severe) | Any value with altered mental/physical state requiring assistance | Medical emergency | 0 events |
Source: International Hypoglycaemia Study Group; Battelino et al., Diabetes Care, 2019
Clinical consequences of frequent hypoglycemia:
- Impaired cognitive function (acute and potentially chronic)
- Cardiac arrhythmias and QT prolongation
- Hypoglycemia unawareness (blunted counter-regulatory responses)
- Increased fall risk (especially older adults)
- Reduced quality of life and diabetes distress
- Potential association with cardiovascular events
Risk factors for increased TBR:
- Adrenal insufficiency or hypopituitarism
- Intensive insulin therapy without CGM or frequent monitoring
- Alcohol consumption
- Irregular meal timing or skipped meals
- Increased physical activity without carbohydrate adjustment
- Renal impairment (reduced insulin clearance)
⚠️ Red Flag: When to Act on Time Below Range
If your Time Below Range (< 70 mg/dL) is consistently above 4% for 2+ weeks:
- Immediate: Review your insulin doses, meal timing, and exercise patterns
- Within 24-48 hours: Contact your diabetes educator or doctor
- Do NOT: Simply eat more to treat frequent lows—this creates a cycle of highs and lows
TBR >4% means you’re spending more than 58 minutes per day in hypoglycemia. This is not just uncomfortable—it’s dangerous and requires professional intervention.
Reading Your Glucose Report
Ambulatory Glucose Profile (AGP)
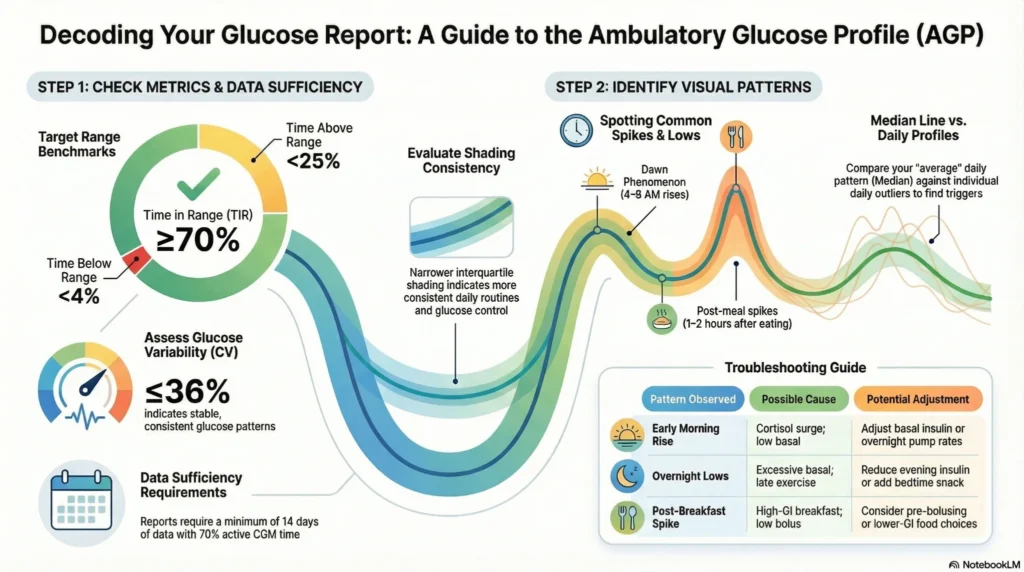
The AGP is a visual summary displaying glucose patterns from CGM data, typically over 14 days.
Core AGP Components:
| Report Section | Information Displayed | How to Interpret |
| Glucose Statistics | Mean glucose, GMI, CV, time in ranges | Overall control summary; compare to targets |
| Median Line (50th percentile) | Typical glucose at each time of day | Your “average” daily pattern |
| Interquartile Range (25th-75th percentile) | Where 50% of readings fall | Narrower shading = more consistent patterns |
| 10th-90th Percentile Range | Where 80% of readings fall | Shows extent of variability |
| Daily Glucose Profiles | Individual days overlaid | Identifies outlier days vs. typical patterns |
| Hourly Statistics | Glucose trends by hour | Reveals dawn phenomenon, post-meal spikes, overnight patterns |
Source: Johnson ML, et al. Utilizing the Ambulatory Glucose Profile to Standardize and Implement Continuous Glucose Monitoring in Clinical Practice. Diabetes Technol Ther. 2019;21(S2):S217-S225.
AGP Interpretation Steps:
- Check time in ranges — Is TIR ≥70%, TBR <4%, TAR <25%?
- Assess variability (CV) — Is it ≤36%?
- Identify patterns — When do highs/lows occur?
- Dawn phenomenon: Rising glucose 4-8 AM
- Post-meal spikes: Elevated 1-2 hours after eating
- Overnight lows: Hypoglycemia during sleep
- Pre-meal drops: Hypoglycemia before scheduled meals
- Evaluate consistency — Is the shaded area narrow (consistent) or wide (erratic)?
- Review daily profiles — Are any days markedly different (sick days, travel, activity)?
Common AGP Patterns and Implications:
| Pattern Observed | Possible Cause | Consideration |
| Early morning rise (dawn phenomenon) | Cortisol surge; insufficient basal insulin | May need basal insulin adjustment or overnight insulin pump rate change |
| Overnight low glucose | Excessive basal insulin; late exercise | Reduce evening long-acting insulin; bedtime snack; review activity timing |
| Post-breakfast spike | Insufficient mealtime insulin; high-GI breakfast | Adjust insulin-to-carb ratio; choose lower-GI foods; consider pre-bolusing |
| Wide interquartile range | High variability; inconsistent routines | Structured meal timing; review insulin dosing; stress management |
| High afternoon/evening glucose | Accumulated carbs; insulin resistance peak; stress | Review afternoon snack choices; adjust dinner insulin; assess cortisol patterns |
Data sufficiency requirements:
- More days (30+) provide greater confidence in pattern identification
- Minimum 14 days of data recommended
- At least 70% CGM active time (equivalent to 10 days of continuous data)
- Fewer days may show incomplete patterns
When to Contact Your Doctor
Emergency guidance and clinical consultation thresholds on this site are established under our medical governance framework.
Certain glucose patterns or metrics require medical consultation rather than self-adjustment.
Urgent Medical Attention Needed:
- Blood glucose >250 mg/dL with ketones present (Type 1 diabetes — risk of diabetic ketoacidosis)
- Blood glucose >400 mg/dL regardless of symptoms
- Blood glucose <54 mg/dL that does not respond to fast-acting carbohydrates within 15 minutes
- Severe hypoglycemia (altered mental status, seizure, loss of consciousness) — call emergency services
- Hyperosmolar hyperglycemic state symptoms: extreme thirst, confusion, visual changes (Type 2 diabetes emergency)
Schedule Provider Consultation:
- HbA1c consistently above target despite medication adherence
- Time in Range <70% over multiple weeks
- Time Below Range >4% (frequent hypoglycemia)
- Coefficient of Variation >36% (high glucose variability)
- Unexplained glucose pattern changes lasting >1 week
- New medications that may affect glucose (steroids, certain psychiatric drugs)
- Pregnancy planning or early pregnancy (require specialized glucose targets)
- Frequent hypoglycemia unawareness (inability to detect lows)
- CGM/meter readings consistently inconsistent with how you feel
Questions to Bring to Your Appointment:
- “My Time in Range is [X]%. What adjustments might improve this?”
- “I notice glucose spikes after [specific meals/times]. What strategies could help?”
- “My HbA1c is [X]%, but my GMI is [Y]%. Why might these differ?”
- “I’m experiencing lows at [specific times]. Should I adjust my [insulin/medication]?”
- “My AGP shows [specific pattern]. What does this suggest about my treatment?”
Educational framing: This page provides educational information about glucose metrics and should not replace professional medical advice. Always consult a qualified healthcare provider for diagnosis, treatment, or personalized glucose management recommendations. Our conflict of interest policy ensures editorial independence.
Educational framing: This page provides educational information about glucose metrics to facilitate informed discussions with your healthcare team. Individual glucose targets and treatment plans must be determined by qualified medical professionals based on your complete health profile. For the scope of our medical reviewer’s authority, see Dr. Das’s credentials and limitations.
How to Discuss These Metrics with Your Doctor
Many people feel intimidated bringing up glucose data with their healthcare provider. Here’s how to prepare for a productive conversation:
What to Bring to Your Appointment:
- Your AGP report (if you use CGM) – most apps generate this automatically under “Reports” or “Share”
- List of specific patterns you’ve noticed – write down 2-3 observations like “I spike to 220 mg/dL within 1 hour of breakfast” or “I drop below 60 mg/dL most afternoons around 3 PM”
- Your current medication regimen – including doses and timing
- Questions about YOUR specific targets – not just general guidelines
Sample Questions That Get Results:
- “My Time in Range is [X]%. What is a realistic target for me given my [age/health conditions]?”
- “I notice my glucose spikes after [specific meal/time]. What adjustment would you recommend—insulin timing, food choices, or medication change?”
- “My HbA1c is [X]%, but my GMI shows [Y]%. Why might these differ, and which should I focus on?”
- “I’m experiencing lows at [specific time]. Should I adjust my [insulin type] dose, or is this a meal timing issue?”
- “My AGP shows [specific pattern—dawn phenomenon, overnight lows, post-lunch spikes]. What does this suggest about my treatment plan?”
How to Advocate for CGM if Your Doctor is Hesitant:
If you believe CGM would help but your doctor hasn’t offered it:
- Present your case with data: “I’m checking 6-8 times daily with fingersticks and still missing patterns. My TBR estimation shows I may be spending 15% of the day hypoglycemic.”
- Reference clinical guidelines: “The ADA Standards of Care recommend CGM for anyone on intensive insulin therapy. I’m taking [X] injections daily.”
- Emphasize hypoglycemia risk: “I’ve had [number] severe lows in the past [timeframe]. CGM alerts could prevent these.”
- Ask about a trial: “Can we try CGM for 2-3 months and evaluate if it improves my control? Some manufacturers offer trial periods.”
If your doctor remains resistant and you believe CGM is medically appropriate, seeking a second opinion from an endocrinologist is reasonable.
What to Do If You Don’t Understand Your Results:
It’s completely normal to feel confused by glucose reports. If your doctor’s explanation doesn’t make sense:
- Ask them to slow down: “Can you explain that again using simpler terms?”
- Request a visual: “Can you show me on the graph what you mean?”
- Repeat back: “So what I’m hearing is [your interpretation]. Is that correct?”
- Get it in writing: “Can you write down my specific targets so I can reference them at home?”
Remember: Understanding your glucose metrics is part of good self-care. There are no “stupid questions”—if something isn’t clear, speak up. Your doctor’s job is to make this understandable for YOU.
Frequently Asked Questions
What is a good Time in Range for Type 2 diabetes?
The target is the same for Type 1 and Type 2 diabetes: >70% of readings between 70-180 mg/dL. This translates to about 17 hours per day in the healthy range. If you’re achieving 60-70%, that’s good progress but worth discussing improvement strategies with your doctor. Below 50% suggests your current treatment plan needs adjustment.
Is 7.5% HbA1c bad?
It depends on your individual circumstances. For most adults with diabetes, the general target is <7.0%, so 7.5% is slightly above goal—not dangerously high, but indicating room for improvement. However, if you’re over 65 with other health conditions or have a history of severe hypoglycemia, 7.5-8.0% may be an appropriate target set by your doctor. The key is comparing your result to YOUR individualized goal, not a universal number.
How much does a CGM cost without insurance?
Expect $150-300 per month depending on the device. Freestyle Libre 3 runs about $150-200/month (most affordable), while Dexcom G7 and Medtronic Guardian cost $240-300/month. Prices include sensors and transmitters. Without insurance, consider manufacturer discount programs—Dexcom offers cash-pay discounts, and Abbott has affordability programs for Libre users.
Can I improve Time in Range without medication changes?
Yes, in many cases. Strategies that can improve TIR by 5-15% without medication changes include:
Eating vegetables and protein before carbohydrates at meals
Taking a 10-15 minute walk after meals
Consistent meal timing (reduces glucose variability)
Choosing lower glycemic index foods (whole grains vs. refined)
Pre-bolusing insulin 15-20 minutes before eating (if you use mealtime insulin)
However, if your TIR is below 50% or your HbA1c is significantly above target, medication adjustment is likely needed for meaningful improvement.
What’s the difference between HbA1c and GMI?
HbA1c is a lab blood test measuring your 2-3 month average glucose by checking how much sugar has attached to red blood cells. GMI (Glucose Management Indicator) is a calculated estimate of what your HbA1c should be based on your CGM’s 14-day average glucose. They usually match within ±0.5%, but GMI updates faster (14 days vs. 3 months) and isn’t affected by red blood cell disorders. Think of GMI as “what your next HbA1c lab will probably show.” The clinical study that established GMI as a standardized metric introduced this term to standardize how CGM data estimates HbA1c equivalents.
How do I know if my glucose monitor is accurate?
CGMs are typically accurate within 8-10% (MARD score). To verify:
Check the CGM reading against a fingerstick when glucose is stable (not rising or falling rapidly)
Expect differences of 10-15 mg/dL—this is normal
If consistently off by >20 mg/dL, calibrate if your model allows, or contact manufacturer
CGMs are less accurate in first 24 hours after insertion and during rapid glucose changes
All FDA-approved CGMs (Dexcom G7, Libre 3, Guardian 4) are approved for treatment decisions without confirmatory fingersticks.
What does it mean if my HbA1c is 7.0% but my GMI is 7.8%?
This discrepancy (GMI higher than lab HbA1c) could mean:
Your glucose control worsened recently (GMI reflects the past 14 days; HbA1c reflects the past 2-3 months)
You’re not wearing your CGM consistently (need >70% wear time for accurate GMI)
You have a condition affecting red blood cells (anemia, hemoglobinopathy) that lowers your HbA1c measurement
If the difference persists, discuss with your doctor. A larger gap suggests your lab HbA1c may not accurately reflect your true glucose control.
Should I check my blood sugar before or after exercise?
Both. Check 15-30 minutes before exercise to avoid starting too low (<100 mg/dL increases hypoglycemia risk during activity). Also check immediately after, and again 2-4 hours later, as delayed hypoglycemia can occur post-exercise. If using CGM, watch for downward trends during activity and consider reducing exercise if glucose is rapidly dropping.
What’s considered dangerously high blood sugar?
Urgent: >250 mg/dL with ketones present (Type 1 diabetes—risk of diabetic ketoacidosis)
Very high: >400 mg/dL regardless of symptoms
Medical emergency: Any blood sugar level with altered mental status, severe confusion, or difficulty breathing
For Type 2 diabetes, sustained readings >300 mg/dL with symptoms (extreme thirst, frequent urination, confusion) may indicate hyperosmolar hyperglycemic state—seek immediate medical care.
How can I reduce my HbA1c naturally?
Lifestyle strategies that typically reduce HbA1c by 0.3-1.0% include:
Losing 5-10% of body weight if overweight
150 minutes per week of moderate aerobic activity (walking, swimming)
Replacing refined carbohydrates with whole grains and vegetables
Eating protein and vegetables before carbohydrates at meals
Getting 7-8 hours of quality sleep nightly
Stress management (cortisol raises blood sugar)
These work best for HbA1c in the 7.0-8.5% range. If your HbA1c is >9%, medication is usually necessary for safe reduction
Does the time of day I check my blood sugar matter?
Yes. Fasting morning glucose (before breakfast) tells you how well your overnight basal insulin or liver glucose production is controlled. Post-meal glucose (1-2 hours after eating) shows your insulin response to food. Most providers want to see both. If you can only check once daily, fasting glucose is usually the priority, but discuss with your doctor based on your specific treatment plan.
What should my blood sugar be at bedtime?
For most adults with diabetes, bedtime glucose of 90-150 mg/dL is a safe target to prevent overnight lows. If you tend to have dawn phenomenon (rising glucose in early morning), your provider may recommend going to bed slightly lower (90-120 mg/dL). If you have hypoglycemia unawareness or are at high risk for nighttime lows, a higher bedtime target (120-180 mg/dL) may be safer.
How often should I check my HbA1c?
Diabetes, meeting targets: Every 6 months
Diabetes, not meeting targets or treatment changes: Every 3 months
Prediabetes: Annually
CGM users: May extend to 6 months if using GMI for interim monitoring
More frequent testing doesn’t improve control—HbA1c reflects 2-3 months of averages, so checking monthly shows little change.
Can stress affect my blood sugar?
Yes, significantly. Stress hormones (cortisol, epinephrine) trigger the liver to release stored glucose, raising blood sugar by 20-100+ mg/dL in some people. Chronic stress also promotes insulin resistance. Stress management techniques (meditation, exercise, adequate sleep) can improve glycemic control. If you notice consistent glucose spikes during stressful periods, discuss stress-related glucose patterns with your provider.
What’s the best app for tracking Time in Range?
Dexcom Clarity (for Dexcom users) – excellent AGP reports
LibreView (for Freestyle Libre users) – simple interface, good pattern detection
CareLink (for Medtronic users) – integrates with insulin pumps
Third-party options include mySugr, Glucose Buddy, and Diabetes:M for manual tracking and comprehensive logging beyond CGM data.
How do I explain my glucose data to my doctor in 5 minutes or less?
Bring these 4 numbers to your appointment:
Your most recent HbA1c or GMI
Your Time in Range percentage (past 14 days)
Your Time Below Range percentage
One specific pattern you’ve noticed (e.g., “I spike above 200 every morning before breakfast” or “I drop below 70 most afternoons around 3 PM”)
If you have a CGM, bring your
References
All cited research is evaluated according to our source verification standards. For methodology transparency, see our data access policy.
Centers for Disease Control and Prevention (CDC). National Diabetes Statistics Report, 2024. Atlanta, GA: Centers for Disease Control and Prevention, US Dept of Health and Human Services; 2024.
American Diabetes Association. Standards of Medical Care in Diabetes—2024. Diabetes Care. 2024;47(Supplement 1):S1-S321. doi:10.2337/dc24-SINT
Battelino T, Danne T, Bergenstal RM, et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations From the International Consensus on Time in Range. Diabetes Care. 2019;42(8):1593-1603. doi:10.2337/dci19-0028
Nathan DM, Kuenen J, Borg R, Zheng H, Schoenfeld D, Heine RJ. Translating the A1C Assay Into Estimated Average Glucose Values. Diabetes Care. 2008;31(8):1473-1478. doi:10.2337/dc08-0545
Bergenstal RM, Beck RW, Close KL, et al. Glucose Management Indicator (GMI): A New Term for Estimating A1C From Continuous Glucose Monitoring. Diabetes Care. 2018;41(11):2275-2280. doi:10.2337/dc18-1581
Danne T, Nimri R, Battelino T, et al. International Consensus on Use of Continuous Glucose Monitoring. Diabetes Care. 2017;40(12):1631-1640. doi:10.2337/dc17-1600
International Hypoglycaemia Study Group. Glucose Concentrations of Less Than 3.0 mmol/L (54 mg/dL) Should Be Reported in Clinical Trials: A Joint Position Statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2017;40(1):155-157. doi:10.2337/dc16-2215
The Diabetes Control and Complications Trial Research Group. The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus. N Engl J Med. 1993;329(14):977-986. doi:10.1056/NEJM199309303291401
Beck RW, Bergenstal RM, Riddlesworth TD, et al. Validation of Time in Range as an Outcome Measure for Diabetes Clinical Trials. Diabetes Care. 2019;42(3):400-405. doi:10.2337/dc18-1444
Johnson ML, Martens TW, Criego AB, Carlson AL, Simonson GD, Bergenstal RM. Utilizing the Ambulatory Glucose Profile to Standardize and Implement Continuous Glucose Monitoring in Clinical Practice. Diabetes Technol Ther. 2019;21(S2):S2-17-S2-25. doi:10.1089/dia.2019.0034
Colberg SR, Sigal RJ, Yardley JE, et al. Physical Activity/Exercise and Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care. 2016;39(11):2065-2079. doi:10.2337/dc16-1728
Shukla AP, Iliescu RG, Thomas CE, Aronne LJ. Food Order Has a Significant Impact on Postprandial Glucose and Insulin Levels. Diabetes Care. 2015;38(7):e98-e99. doi:10.2337/dc15-0429
International Diabetes Federation. Guideline for Management of PostMeal Glucose in Diabetes. 2011.
National Institute for Health and Care Excellence (NICE). Type 1 Diabetes in Adults: Diagnosis and Management. NICE Guideline NG17. Updated 2022.
American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 201: Pregestational Diabetes Mellitus. Obstet Gynecol. 2018;132(6):e228-e248.
Page last updated: March 24, 2026
Medical review: Dr. Rishav Das, M.B.B.S. — March 24, 2026 [Review process]
This page provides educational information about glucose metrics and should not replace professional medical advice. Always consult a qualified healthcare provider for diagnosis, treatment, or personalized glucose management recommendations.
Contact our medical team : contact@wearablewellnessguide.com
