Evidence-Based Guide to Selecting Glucose Monitors In 2026
Compare CGM, Flash, and traditional glucose monitors & meters. Explore accuracy standards, costs, and clinical use cases for diabetes management.
Written by Dr. Rishav Das, M.B.B.S. -Wellness Device Data Analyst
| Consumer Device Accuracy Specialist
Medically reviewed according to the medical standards outlined on our About page
Introductory Context
If you’re researching glucose monitors, you may be newly diagnosed with diabetes, helping a family member manage their condition, or looking to improve control after years of traditional testing. Whatever brings you here, choosing the right monitoring approach can feel overwhelming with so many options available.
Glucose monitoring technology has evolved significantly. Today’s options range from simple meters requiring finger pricks to advanced continuous monitors that track glucose automatically throughout the day and night. Understanding the differences between these technologies, their costs, and which situations each serves best helps you make an informed choice aligned with your health needs, lifestyle, and budget.
Who This Guide Serves:
- Newly diagnosed individuals learning about monitoring options for the first time
- Parents of children with diabetes seeking less invasive or remote monitoring solutions
- Long-time diabetes managers considering upgrades from traditional testing
- Active individuals and athletes needing real-time glucose awareness during exercise
- Caregivers and family members helping loved ones select appropriate devices
This guide provides evidence-based information on glucose monitoring devices, their mechanisms, and selection considerations. It complements—but does not replace—professional medical advice. Always consult your healthcare provider before selecting or changing glucose monitoring methods, as individual needs vary based on diabetes type, treatment regimen, and personal health factors.
All devices reviewed on this page were purchased at market price with no manufacturer funding or affiliate relationships, in accordance with our conflict of interest policy.
Quick Match: Find Your Device Type in 60 Seconds
Choose your starting point based on your situation:
🆕 Just Diagnosed or New to Glucose Monitoring?
→ Start with Traditional Blood Glucose Meters section to understand basic monitoring, then explore continuous options if your healthcare provider recommends them.
👨👩👧 Parent Monitoring a Child with Diabetes?
→ Jump to Continuous Glucose Monitors (CGMs) section—specifically Dexcom G7 or FreeStyle Libre 3, which offer remote monitoring features allowing you to track your child’s glucose from your smartphone.
🏃 Active Lifestyle or Athlete?
→ See Continuous Glucose Monitors (CGMs) section for devices providing real-time glucose awareness during exercise without interrupting activity. Look for water-resistant options rated for swimming and high-intensity workouts.
💰 Budget-Conscious or Concerned About Costs?
→ Review both Traditional Blood Glucose Meters (lowest ongoing cost) and Cost Considerations section to understand insurance coverage options and patient assistance programs that may reduce CGM expenses significantly.
💉 Use Insulin Multiple Times Daily (Type 1 or Intensive Type 2)?
→ Continuous Glucose Monitors (CGMs) section—these devices have the strongest evidence base for insulin users and typically receive insurance coverage more readily for this indication.
📊 Want to See Glucose Patterns and Trends?
→ Explore both Continuous Glucose Monitors and Flash Glucose Monitors sections to understand how these technologies reveal patterns traditional meters cannot show.
Not sure which applies to you? Continue reading from the beginning—this guide walks through all options systematically, helping you identify the best fit as you learn.
Why People Switch to Continuous Glucose Monitoring
Many individuals with diabetes face daily challenges that traditional testing methods struggle to address:
Sleep Disruption and Nighttime Worry
Waking up multiple times at night to check blood sugar levels disrupts sleep quality for both patients and caregivers. Parents of children with diabetes often set alarms every 2-3 hours, leading to chronic sleep deprivation and anxiety about dangerous overnight lows.
Finger-Prick Fatigue
Testing 8-10 times daily can make fingertips sore, calloused, and painful. Many people report difficulty with work tasks requiring fine motor skills, such as typing or handling small objects, due to tender fingertips.
Public Testing Concerns
Drawing blood in public settings—at restaurants, during meetings, or while traveling—can feel uncomfortable or inconvenient. Some individuals delay necessary tests to avoid these situations, compromising glucose control.
Missing Critical Glucose Changes
Traditional meters provide single-point measurements, which can miss rapid glucose drops or spikes occurring between tests. This creates uncertainty about whether glucose levels are stable or changing dangerously.
Data Overwhelm Without Context
Individual glucose readings don’t show trends or patterns. Without seeing how food, activity, stress, and medication interact over time, making adjustments becomes guesswork rather than informed decision-making.
Continuous glucose monitoring technologies were developed to address these specific challenges, offering different approaches to reduce testing burden while improving glucose awareness.
- Evidence-Based Guide to Selecting Glucose Monitors In 2026
- Introductory Context
- Quick Match: Find Your Device Type in 60 Seconds
- Why People Switch to Continuous Glucose Monitoring
- Types of Glucose Monitors
- Continuous Glucose Monitors (CGMs): Best for Insulin Users and Intensive Management
- Flash Glucose Monitors: Budget-Friendly Alternative with Trend Data
- Traditional Blood Glucose Meters: Affordable and Reliable Testing
- Side-by-Side Device Comparison
- Top-Rated Glucose Monitors by Category (2026)
- Non-Invasive Glucose Monitors (Emerging)
- How Each Technology Works
- Device Selection Considerations
- Advanced Monitoring Features
- Understanding Glucose Metrics
- Frequently Asked Questions About Glucose Monitors
- How painful are CGM sensors when inserted?
- Can I shower, swim, or exercise with a CGM?
- How long does insurance approval take for a CGM?
- What if my CGM sensor fails or gives inaccurate readings?
- Do I need a smartphone to use a CGM?
- How accurate are CGMs compared to fingerstick meters?
- Will my insurance cover a CGM?
- How often do I need to replace CGM sensors?
- Can I use a CGM if I'm pregnant or planning pregnancy?
- Data Management and Sharing
- Connect with the Glucose Monitoring Community
- When to Consult a Healthcare Provider
- References
Types of Glucose Monitors
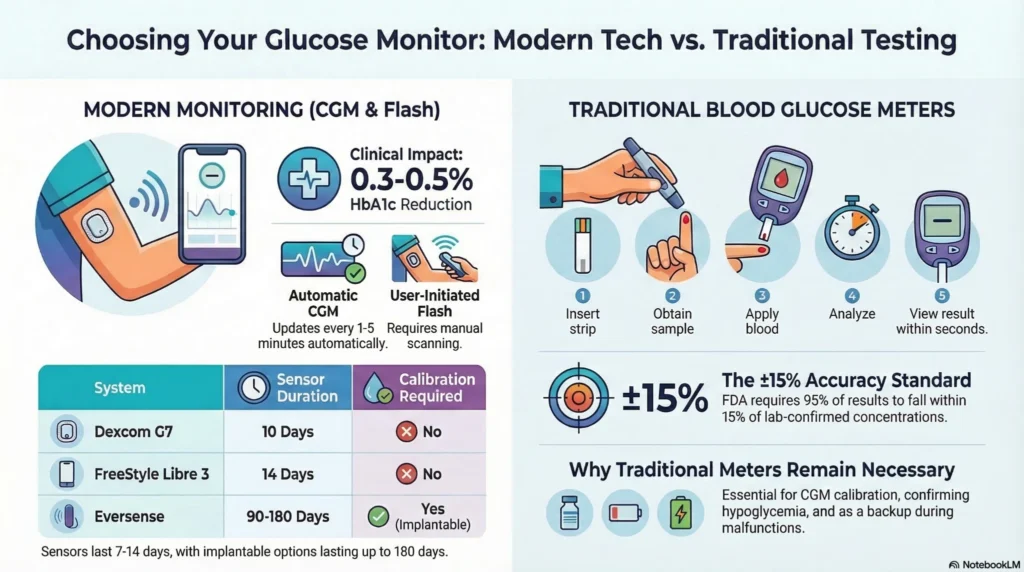
Continuous Glucose Monitors (CGMs): Best for Insulin Users and Intensive Management
Device Characteristics
| Feature | Description |
| Measurement Method | Subcutaneous sensor measures interstitial fluid glucose |
| Reading Frequency | Every 1-5 minutes (automatic) |
| Sensor Duration | 7-14 days (varies by model) |
| Display Method | Dedicated receiver or smartphone app |
| Calibration | Most modern systems require no fingerstick calibration |
| Alerts | Real-time high/low glucose alerts available |
Key CGM Systems (Current Market)
- Eversense: 90-180 day implantable sensor (requires minor procedure)
- Dexcom G7: 10-day sensor, 5-minute readings, no calibration required
- FreeStyle Libre 3: 14-day sensor, 1-minute readings, no calibration required
- Medtronic Guardian Connect: 7-day sensor, may require calibration
Life-Changing Benefits of Continuous Monitoring
Beyond clinical improvements, CGM users report significant lifestyle enhancements:
Sleep Through the Night with Confidence
Programmable alerts notify users before glucose reaches dangerous levels, allowing uninterrupted sleep while maintaining safety. Parents can monitor children remotely through smartphone apps, reducing nighttime anxiety without constant physical checks.See Food Impact in Real-Time
Watch how different meals affect glucose levels within minutes rather than waiting hours to test. This immediate feedback helps identify problem foods and portion sizes, making dietary adjustments more intuitive and effective.Exercise Without Guesswork
Check glucose levels before, during, and after physical activity without interrupting workouts for finger pricks. Athletes and active individuals can identify patterns showing when they need pre-exercise snacks or post-workout adjustments.Dramatically Fewer Finger Pricks
Modern CGM systems like Dexcom G7 and FreeStyle Libre 3 require zero fingerstick calibrations. Users who previously tested 8-10 times daily often reduce to zero routine finger pricks, reserving traditional meters only for occasional confirmation when needed.Better Long-Term Control
Clinical research demonstrates CGM users typically achieve 0.3-0.5% HbA1c improvements compared to traditional monitoring. This reduction translates to meaningful decreases in long-term complication risks, including neuropathy, retinopathy, and cardiovascular disease.Early Warning of Dangerous Trends
Trend arrows and predictive alerts warn users when glucose is dropping rapidly or rising persistently—not just when it crosses a threshold. This advance notice enables preventive action rather than reactive crisis management.
Evidence Base
Research indicates CGMs may improve glycemic control in individuals with type 1 diabetes, with studies showing reductions in HbA1c of 0.3-0.5% compared to traditional monitoring.[1,2] Benefits in type 2 diabetes populations are emerging, with evidence suggesting improved glucose time-in-range.[3]
⚡ TL;DR: CGMs automatically measure glucose every 1-5 minutes via a small sensor worn on your arm or abdomen for 7-14 days. They send real-time readings and alerts to your smartphone, eliminating most finger pricks. Cost: $200-400/month without insurance, $35-100/month with coverage. Best for insulin users, those with hypoglycemia unawareness, or anyone wanting continuous glucose awareness and trend data. Most insurance plans cover for Type 1 diabetes; Type 2 coverage requires intensive insulin therapy typically.
Ready to Move Forward?
✓ Discuss with your doctor: Bring this guide to your next appointment to discuss which device type fits your treatment plan
✓ Check insurance coverage: Call your insurance to verify CGM/meter coverage and copay amounts
✓ Explore manufacturer resources: Visit [device name] manufacturer website for detailed specifications and patient support programs
Flash Glucose Monitors: Budget-Friendly Alternative with Trend Data
Technology Overview
| Characteristic | Flash Glucose Monitor Specifics |
| Reading Trigger | User-initiated scan (not automatic) |
| Sensor Type | Subcutaneous filament in interstitial fluid |
| Data Storage | Sensor stores 8 hours of glucose data |
| Scan Requirement | Must scan at least every 8 hours to maintain data continuity |
| Alert Capability | Limited or app-dependent (varies by model) |
| Primary Example | FreeStyle Libre 2 (scan-based with optional alerts) |
Key Distinctions from CGMs
- Cost considerations: May be lower-cost alternative to full CGM systems
- User-initiated readings: Requires active scanning rather than automatic transmission
- Alert limitations: May not provide comprehensive real-time alerts
- Trend data: Available when scanned, showing glucose direction and rate of change
Clinical Context
Flash glucose monitoring has demonstrated improved glucose control compared to traditional fingerstick testing in some populations, though evidence is most robust for intensive insulin therapy users.[4]
⚡ TL;DR: Flash monitors use the same sensor technology as CGMs but require you to scan the sensor with your phone to see readings (not automatic). Sensors last 14 days. Cost: $100-150/month without insurance, $25-75/month with coverage. Provides trend graphs and directional arrows but limited automatic alerts. Best for budget-conscious users wanting more data than fingersticks provide, or Type 2 diabetes patients not requiring intensive alarm features.
Ready to Move Forward?
✓ Discuss with your doctor: Bring this guide to your next appointment to discuss which device type fits your treatment plan
✓ Check insurance coverage: Call your insurance to verify CGM/meter coverage and copay amounts
✓ Explore manufacturer resources: Visit [device name] manufacturer website for detailed specifications and patient support programs
Traditional Blood Glucose Meters: Affordable and Reliable Testing
Measurement Process
Step-by-Step Operation:
- Insert test strip into meter
- Obtain blood sample via fingerstick lancet
- Apply blood drop to test strip
- Meter analyzes blood glucose via enzymatic reaction
- Result displays in 5-10 seconds
Meter Categories
| Meter Type | Features | Use Cases |
| Basic Meters | Simple readout, minimal features | Infrequent testing, budget constraints |
| Advanced Meters | Bluetooth connectivity, trend tracking, averaging | Regular monitoring, data tracking needs |
| Talking Meters | Audio readout of results | Visual impairment accommodation |
| Multi-test Meters | Ketone testing capability | Type 1 diabetes, DKA risk management |
Accuracy Standards
FDA requires blood glucose meters to meet ISO 15197:2013 standards: 95% of results must fall within ±15 mg/dL (±0.8 mmol/L) at glucose concentrations <100 mg/dL or ±15% at ≥100 mg/dL.[5]
When Traditional Meters Remain Necessary
- Backup when CGM is unavailable or malfunctioning
- CGM calibration (when required by specific systems)
- Confirmation of hypoglycemia before treatment decisions
- Situations requiring immediate, confirmed blood glucose value
⚡ TL;DR: Traditional meters require finger-prick blood samples and provide one reading per test. No ongoing sensor costs—just test strips ($20-100/month for typical testing frequency). Most accurate method for single-point readings. Best for infrequent testing needs, budget constraints, backup monitoring, or situations requiring confirmed blood glucose values. Universally covered by insurance with minimal copays.
Ready to Move Forward?
✓ Discuss with your doctor: Bring this guide to your next appointment to discuss which device type fits your treatment plan
✓ Check insurance coverage: Call your insurance to verify CGM/meter coverage and copay amounts
✓ Explore manufacturer resources: Visit [device name] manufacturer website for detailed specifications and patient support programs
Side-by-Side Device Comparison
Compare key features to identify which monitoring approach best fits your needs:
| Feature | Traditional Meter | Flash Monitor | Continuous CGM |
|---|---|---|---|
| How It Works | Finger-prick blood sample | Scan sensor on arm | Automatic continuous readings |
| Reading Frequency | When you test (typically 4-8x daily) | When you scan (scan at least every 8 hours) | Every 1-5 minutes automatically |
| Sensor Wear Time | N/A (new strip each test) | 14 days | 7-14 days depending on model |
| Alerts for Highs/Lows | No (you must test to know) | Limited or app-dependent | Yes, real-time customizable alerts |
| Trend Information | No | Yes, when you scan | Yes, continuous with predictive arrows |
| Monthly Cost (Without Insurance) | $20-100 (test strips) | $100-150 (sensors) | $200-400 (sensors + transmitter) |
| Monthly Cost (With Insurance) | $0-30 copay | $25-75 copay | $35-100 copay (varies widely) |
| FDA Accuracy (MARD) | ±15% at >100 mg/dL | 9-10% MARD | 8-10% MARD |
| Calibration Required | N/A | No (most models) | No (most modern systems) |
| Best For | Budget-conscious users, infrequent testing needs, backup monitoring | Active Type 2, cost-conscious seekers of trend data | Insulin users, hypoglycemia unawareness, intensive management |
| Medicare Coverage | Yes | Limited (coverage expanded recently) | Yes, with qualifying criteria |
| Water Resistance | Meter must stay dry | Most sensors: showering OK, swimming OK | Most sensors: showering OK, swimming OK |
| Learning Curve | Minimal (5-10 minutes) | Low (15-30 minutes) | Moderate (1-2 hours initial setup) |
| Smartphone Integration | Some advanced models | Yes (most modern versions) | Yes (required for most systems) |
Quick Decision Guide Based on This Table:
- Choose Traditional Meters if: You test infrequently (1-2x daily), have budget constraints, or don’t need trend information
- Choose Flash Monitors if: You want trends without automatic alerts, are cost-conscious but need more data than meters provide, or have Type 2 diabetes not requiring intensive insulin
- Choose Continuous CGMs if: You use insulin multiple times daily, experience hypoglycemia unawareness, want predictive alerts, or need remote monitoring capability for children/dependents
Note: Prices shown are approximate 2024 ranges and vary by region, insurance plan, and specific device model. Always verify current costs with manufacturers and your insurance provider.
Top-Rated Glucose Monitors by Category (2026)
Based on clinical accuracy data, user ratings, and evidence-based outcomes:
Best Overall Continuous Glucose Monitor: Dexcom G7
- Why It Leads: 10-day sensor life, 8.1% MARD accuracy, most comprehensive alert system, shortest warm-up time (30 minutes), extensive insurance coverage
- Best For: Type 1 diabetes, insulin pump users, individuals prone to hypoglycemia, parents monitoring children remotely
- Typical Cost: $350-400/month without insurance; $35-100/month with insurance coverage
- User Advantage: No calibration required, real-time sharing with up to 10 followers, integration with insulin pumps
Best Budget CGM Option: FreeStyle Libre 3
- Why It Stands Out: 14-day sensor life (longest available), 9.2% MARD accuracy, lowest monthly CGM cost, 1-minute reading frequency
- Best For: Type 2 diabetes, cost-conscious users seeking continuous monitoring, those not requiring extensive alert customization
- Typical Cost: $200-300/month without insurance; $25-75/month with insurance
- User Advantage: Smallest sensor available, simple application process, growing insurance acceptance
Best Traditional Meter for Accuracy: Contour Next One
- Why Clinicians Recommend It: Second-chance sampling (add more blood to same strip if needed), proven accuracy across wide hematocrit ranges, smartphone connectivity with free app
- Best For: Individuals requiring fingerstick confirmation, those preferring traditional testing, budget-conscious users
- Typical Cost: $20-40/month for strips (testing 4x daily); meter often free
- User Advantage: No coding required, works with generic test strips, results in 5 seconds
Easiest to Use for Seniors and Beginners: FreeStyle Libre 2
- Why It’s Simple: One-button application, large display with clear numbers, optional alarms for safety, no smartphone required (receiver available)
- Best For: Older adults, those uncomfortable with technology, caregivers managing loved ones’ diabetes
- Typical Cost: $150-250/month without insurance; $35-75/month with Medicare or insurance
- User Advantage: Scan-based reading (no automatic alerts unless desired), 14-day sensor, simpler than full CGM systems
Best for Athletes and Active Lifestyles: Dexcom G7
- Why Athletes Choose It: IPX8 water resistance (swim-safe up to 8 feet for 24 hours), withstands high-intensity workouts, provides real-time glucose during exercise without interruption
- Best For: Runners, cyclists, swimmers, gym enthusiasts, outdoor workers
- Typical Cost: Same as Best Overall (above)
- User Advantage: Adhesive designed for movement and sweat, trend arrows show glucose direction during activity
Important Note: “Best” device varies by individual needs, insurance coverage, lifestyle, and diabetes type. These recommendations represent common user preferences and clinical evidence but should be discussed with your healthcare provider to ensure the choice aligns with your specific medical situation.
Next Steps Toward Purchase:
✓ Verify current prices: Device costs change frequently—check manufacturer websites or call DME suppliers for latest pricing
✓ Confirm insurance coverage: Contact your insurance provider with specific device model names to verify coverage
✓ Schedule diabetes educator appointment: Many insurers require diabetes education before approving CGM coverage
✓ Request trial if available: Some endocrinologists have 14-30 day sample sensors from manufacturers for trial before commitment
Non-Invasive Glucose Monitors (Emerging)
Technology Status
| Technology Approach | Development Stage | Key Challenges |
| Optical sensors | Research/early trials | Accuracy in diverse skin types and conditions |
| Electromagnetic | Prototype phase | Interference from body composition variations |
| Transdermal | Limited commercial availability | Reliability across glucose ranges |
| Spectroscopy | Academic research | Calibration stability over time |
Evidence Limitations
- Current non-invasive glucose monitoring technologies face significant validation challenges:
- Environmental interference potential
- Lack of FDA approval for primary glucose monitoring decisions
- Limited peer-reviewed clinical validation data
- Accuracy concerns across physiological variations
Medical Advisory
As of current evidence, non-invasive glucose monitors are not recommended as replacements for established monitoring methods for diabetes management decisions. Individuals should consult healthcare providers before using experimental or non-validated devices.[6]
Ready to Move Forward?
✓ Discuss with your doctor: Bring this guide to your next appointment to discuss which device type fits your treatment plan
✓ Check insurance coverage: Call your insurance to verify CGM/meter coverage and copay amounts
✓ Explore manufacturer resources: Visit [device name] manufacturer website for detailed specifications and patient support programs
How Each Technology Works
CGM Sensor Technology
Mechanism of Action
| Component | Function | Technical Detail |
| Glucose Oxidase Enzyme | Catalyzes glucose reaction | Produces electrical signal proportional to glucose concentration |
| Electrode | Detects enzymatic reaction | Converts chemical signal to electrical current |
| Transmitter | Processes and sends data | Wireless transmission to receiver/smartphone |
| Interstitial Fluid | Measurement medium | Glucose correlates with blood glucose (5-10 minute physiological lag) |
Physiological Lag Explanation
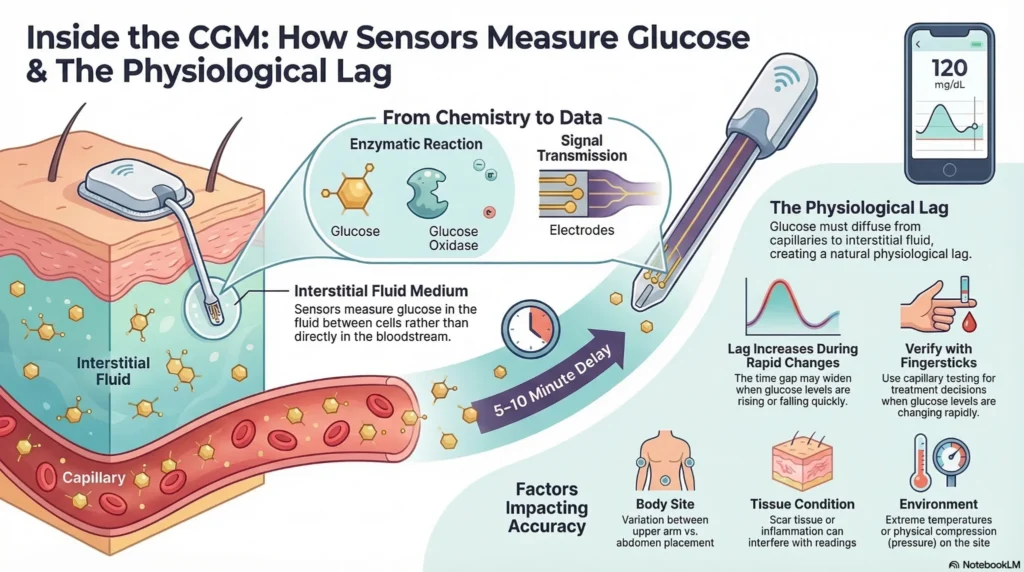
- Blood glucose changes first
- Glucose diffuses from capillaries to interstitial fluid
- CGM sensor detects interstitial glucose
- Reading appears on device 5-10 minutes after blood glucose change
This lag is clinically relevant during rapid glucose fluctuations. During stable periods, interstitial glucose closely mirrors blood glucose.
Sensor Lifespan and Replacement
CGM sensors have defined wear periods due to:
- Enzymatic activity degradation over time
- Tissue response at insertion site
- Foreign body reaction reducing sensor contact
- Manufacturer-validated accuracy windows
💬 Common Concern: “I’m worried about sensor failures during important moments—what if it stops working when I really need it?”
While sensor failures are uncommon (<2% monthly failure rate), smart users stay prepared:
Built-in safeguards:
- Systems alert you 12-24 hours before scheduled sensor expiration
- Low battery warnings give 1-4 hours notice before transmitter shutdown
- Most sensors give error alerts when experiencing technical issues
Your safety backup:
- Always keep: A traditional blood glucose meter with test strips—even long-time CGM users maintain backup meters
- Manufacturer support: All major brands replace faulty sensors free when you contact their 24/7 technical support hotline
- Treatment decisions: FDA recommends fingerstick confirmation before making treatment decisions during suspected sensor inaccuracy anyway
What experienced users do: Keep a small kit with backup meter, lancets, and strips in your bag or car. Most never need it, but the preparedness eliminates the worry.
Wearing sensors beyond approved duration may compromise accuracy and is not recommended.
💬 Common Concern: “I’m worried the technology will be too complicated for me to learn.”
Modern CGMs work with your smartphone—if you can send text messages or check email on your phone, you can use a CGM. The apps are designed for ease of use with large, clear displays showing your glucose number, trend arrow, and graph.
What makes it easier than you think:
- Initial setup walks you through each step with pictures
- Manufacturers provide 24/7 phone support and training videos
- Your diabetes educator can help during your first sensor application
- After the first sensor, most users complete the process in under 2 minutes
Real user perspective: “I’m 68 and thought I was too old to learn new technology. My granddaughter helped me set up the first sensor, and now I do it myself every two weeks with no problem. The peace of mind is worth the initial learning.” —Medicare CGM user
Flash Monitor Scanning Process
How On-Demand Scanning Works
| Step | Process | Data Captured |
| 1. Sensor Storage | Glucose readings stored in sensor memory | Up to 8 hours of continuous data |
| 2. User Initiates Scan | Smartphone/reader placed near sensor | NFC communication activated |
| 3. Data Transfer | Sensor transmits stored glucose readings | Current reading + historical trend |
| 4. Display | Graph shows glucose trajectory | Direction arrows indicate glucose trend |
Scan Frequency Requirements
Flash monitors require scans at least every 8 hours to maintain continuous data. Longer intervals create data gaps. More frequent scanning provides more complete glucose patterns.
Alert Capabilities
Some flash systems offer optional glucose alerts through smartphone apps. Alert functionality varies by model and may require specific app configurations.
Traditional Meter Chemistry
Enzymatic Glucose Measurement
Blood glucose meters use one of two enzymatic reactions:
Glucose Oxidase Method:
- Enzyme reacts with glucose and oxygen
- Produces hydrogen peroxide
- Electrical current measured proportional to glucose
Glucose Dehydrogenase Method:
- Enzyme reacts with glucose
- Produces electrons directly
- Less oxygen-dependent than oxidase method
Both methods convert glucose concentration into electrical signals measured in 5-10 seconds.
Factors Affecting Meter Accuracy
- Blood sample size and application technique
- Hematocrit (red blood cell concentration)
- Altitude and oxygen levels
- Medication interference (some vitamins, acetaminophen)
- Test strip storage and expiration
Device Selection Considerations
Clinical Use Cases
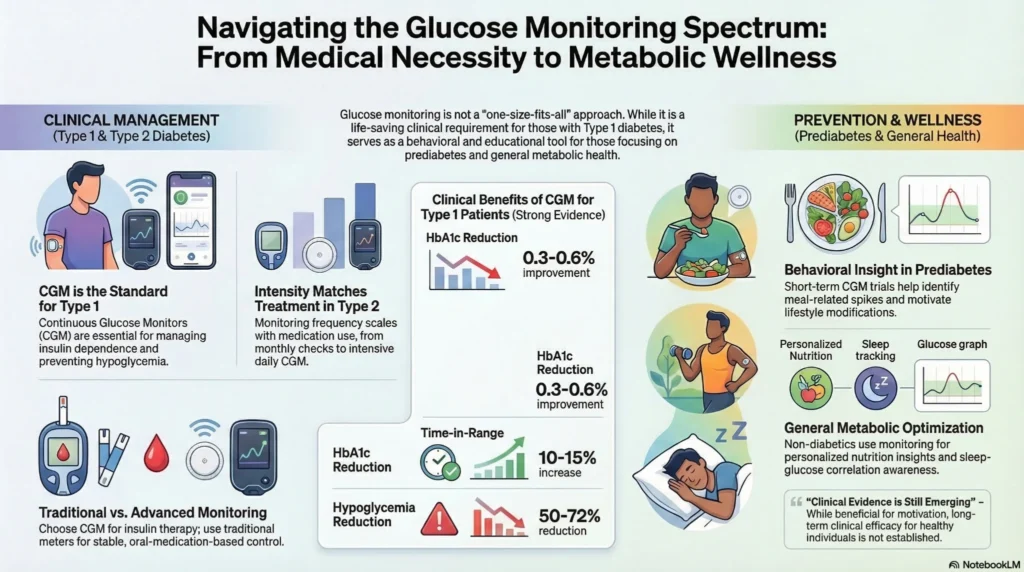
Type 1 Diabetes
Continuous monitoring is generally recommended due to:
- Insulin dosing requires frequent glucose checks
- Hypoglycemia risk management
- Exercise and activity planning
- Overnight glucose monitoring
CGM systems with alerts may reduce severe hypoglycemia episodes and improve time-in-range.
Type 2 Diabetes (Insulin-Treated)
Device selection depends on insulin regimen:
- Basal-bolus insulin: CGM or flash monitoring recommended
- Basal insulin only: Flash monitoring or traditional meters may be sufficient
- Mixed insulin: Consider CGM for optimization
Evidence for CGM benefit in type 2 diabetes is strongest for those on intensive insulin therapy.[3]
Type 2 Diabetes (Non-Insulin)
Traditional meters may be adequate for many individuals. Flash monitoring or CGM may benefit:
- Those adjusting oral medications
- Individuals with variable glucose patterns
- People seeking lifestyle intervention feedback
Consult healthcare providers about monitoring frequency appropriate for treatment plan.
Gestational Diabetes
Traditional meter testing remains standard care in most guidelines. CGM may be used in select cases under medical supervision. Testing frequency and targets should be determined by healthcare providers.
Prediabetes and Prevention
FDA-approved glucose monitors are prescription devices for diabetes management. Consumer CGM programs (non-prescription) exist but should not replace medical evaluation.
Individuals with prediabetes should discuss monitoring approaches with healthcare providers rather than self-initiating continuous monitoring.
Coverage and Cost Factors
Insurance Coverage Criteria
Most health insurance plans cover glucose monitoring with specific requirements:
Medicare Coverage (as of 2023):[9]
- CGM covered for insulin-treated diabetes (type 1 or 2)
- Requires 4+ fingerstick checks per day prior to CGM approval
- Prescription required with specific documentation
- Traditional meter supplies covered for all diabetes types
Private Insurance:
- Coverage varies by plan and state
- Prior authorization often required for CGM
- Step therapy may require traditional meter use first
- Annual out-of-pocket costs depend on deductibles and copays
Cost Without Insurance
Approximate monthly costs (2024):
- Traditional meters: $20-50 (meter) + $50-150 (test strips)
- Flash monitors: $75-150 (sensors only)
- CGM systems: $200-400 (sensors + transmitter)
Prices vary by manufacturer, pharmacy, and quantity purchased.
Consumer CGM Programs
- Non-prescription CGM programs have emerged for wellness use. These differ from prescription diabetes monitoring:
- Should not replace medical monitoring when clinically indicated
- Not FDA-approved for diabetes management decisions
- Positioned as general wellness devices
- May not be covered by insurance
How to Get Insurance Approval for CGM: Step-by-Step Process
Most insurance denials result from incomplete documentation, not lack of medical need. Follow this systematic approach:
Step 1: Verify Coverage (Before Requesting)
- Call your insurance company’s member services with your policy number
- Ask specifically: “Does my plan cover continuous glucose monitors? What are the medical criteria?”
- Request the prior authorization form and coverage policy document
- Note the representative’s name, date, and reference number
Step 2: Gather Required Documentation
Most insurers require:
- ✓ Prescription from your healthcare provider specifying CGM need
- ✓ Diabetes diagnosis documentation (ICD-10 code)
- ✓ Current medication list showing insulin therapy (if applicable)
- ✓ Recent A1C lab results
- ✓ Documentation of:
- Hypoglycemia frequency (if relevant)
- Current monitoring frequency (fingerstick logs)
- Failed attempts to achieve control with current methods
Step 3: Healthcare Provider Letter of Medical Necessity
Your doctor’s letter should include specific phrases insurers look for:
- “Medically necessary for diabetes management”
- Frequency of hypoglycemic episodes (if applicable)
- Intensive insulin therapy regimen details
- How CGM will improve clinical outcomes
- Previous glucose monitoring methods tried
- Current A1C and target goals
Step 4: Submit Complete Prior Authorization
- Submit all documents together (incomplete submissions cause delays)
- Keep copies of everything submitted
- Note submission date and confirmation number
- Typical review timeline: 3-10 business days
Step 5: Follow Up Proactively
- Call insurance every 3-5 days for status updates
- Document all conversations (date, representative name, notes)
- If approaching day 10, escalate to supervisor
If Denied: The Appeal Process
First Level Appeal (Success Rate: 78%)
- Request written denial with specific reason codes
- Review denial reason carefully—often it’s missing documentation
- Obtain additional supporting documents addressing denial reason
- Resubmit with stronger physician letter specifically countering denial rationale
- Cite insurance policy language showing you meet criteria
- Submit within appeal deadline (typically 30-60 days from denial)
Common denial reasons and how to overcome them:
- “Not medically necessary” → Provide hypoglycemia log, A1C progression showing poor control, physician letter emphasizing clinical need
- “Doesn’t meet criteria for insulin therapy” → Submit detailed insulin regimen, prescription records, pharmacy fill history
- “Traditional monitoring is sufficient” → Document fingerstick frequency burden, missed hypoglycemic episodes, quality of life impact statement
Second Level Appeal (External Review)
If first appeal denied:
- Request external review by independent medical reviewer
- Provide all previous documentation plus any new clinical evidence
- External reviewers often overturn insurer decisions
- This process is free—required by law
Getting Professional Help
- Diabetes educators: Many help with prior authorization paperwork
- Manufacturer support programs: Dexcom, Abbott, and Medtronic have reimbursement specialists who assist with appeals
- Patient advocates: Some healthcare systems have insurance navigation specialists
Typical Full Timeline:
- Initial submission to approval: 1-3 weeks
- Denial to first appeal decision: 2-4 weeks
- Second appeal/external review: 4-8 weeks
While Waiting for Approval:
Continue traditional monitoring to document ongoing need
Ask your endocrinologist about manufacturer sample sensors for trial use
Some DME suppliers offer sensors at reduced self-pay rates pending approval
💬 Common Concern: “What if my insurance denies coverage?”
Initial insurance denials are common, but 78% are overturned on appeal. Here’s what helps:
Documentation that strengthens appeals:
- Letter from your healthcare provider explaining medical necessity
- Diabetes diagnosis confirmation and current treatment plan
- Log showing frequent hypoglycemia or poor glucose control with current monitoring
- Previous trials with intensive insulin therapy or documented A1C above target
If appeals don’t succeed:
- Manufacturer patient assistance programs can reduce CGM costs to $0-$75/month for qualifying individuals based on income
- Dexcom Warrior Program: Income-based assistance
- Abbott MyFreeStyle: Patient support program
- Sample/trial programs: Some endocrinologists have manufacturer samples for 14-30 day trials
Alternative: Consider starting with a Flash Glucose Monitor (FreeStyle Libre), which costs less than full CGMs and has broader insurance acceptance for Type 2 diabetes.
Accuracy and Reliability
CGM Accuracy Metrics
Modern CGM systems report Mean Absolute Relative Difference (MARD):
- MARD <10%: Considered clinically accurate
- Dexcom G7: ~8% MARD[7]
- FreeStyle Libre 3: ~7.9% MARD[8]
Lower MARD indicates closer agreement with laboratory glucose measurements.
Accuracy Variability Factors
CGM accuracy may vary with:
- Glucose range (less accurate at very low or very high values)
- Rate of glucose change (rapid changes increase lag effect)
- Sensor placement and insertion quality
- Individual physiological factors
- Days of sensor wear (some drift over time)
When to Confirm with Fingerstick
- CGM/flash monitor readings should be confirmed with traditional meter when:
- During rapid glucose changes
- Glucose alerts indicate severe hypoglycemia
- Symptoms don’t match CGM reading
- Before making treatment decisions about insulin dosing
- CGM shows “?” or error indicators
Lifestyle and Practical Factors
Physical Activity Considerations
Different devices have varying water resistance and durability:
- Most CGM sensors are water-resistant (showering, swimming approved)
- Contact sports may dislodge sensors
- Adhesive performance varies with sweating
- Some systems offer overwraps for additional security
Travel and Time Zones
- CGM receivers/apps adjust to local time zones
- Sensor calibration schedules may need adjustment
- Carry backup supplies and prescription documentation
- Airport security screening generally safe for devices
Skin Sensitivity
Adhesive allergies or skin reactions occur in some users:
- Barrier films available for sensitive skin
- Alternative insertion sites may reduce irritation
- Persistent skin reactions should be discussed with healthcare providers
- Some individuals cannot tolerate adhesive sensors
Device Connectivity
- Many systems offer smartphone integration:
- Compatibility varies by phone operating system
- Data sharing with family members or healthcare providers
- Cloud storage of glucose records
- Integration with insulin pumps (hybrid closed-loop systems)
Advanced Monitoring Features
Hybrid Closed-Loop Systems
Automated Insulin Delivery Integration
Some CGM systems integrate with insulin pumps to create hybrid closed-loop systems:
| System | CGM Component | Insulin Pump | Automation Level |
| Tandem Control-IQ | Dexcom G6/G7 | Tandem t:slim X2 | Adjusts basal insulin automatically |
| Medtronic 780G | Guardian 4 | MiniMed 780G | Adjusts basal and correction boluses |
| Omnipod 5 | Dexcom G6 | Omnipod 5 | Adjusts basal insulin automatically |
How Hybrid Systems Work
- CGM measures glucose every 5 minutes
- Algorithm predicts glucose trajectory
- Insulin pump adjusts insulin delivery automatically
- User still enters carbohydrates and approves meal boluses
These systems reduce manual diabetes management burden while requiring user input for meals.
Evidence for Hybrid Systems
Studies show hybrid closed-loop systems improve time-in-range compared to traditional pump therapy, with reductions in hypoglycemia.[12] Users should receive comprehensive training before initiation.
Glucose Trend Analysis
Rate-of-Change Arrows
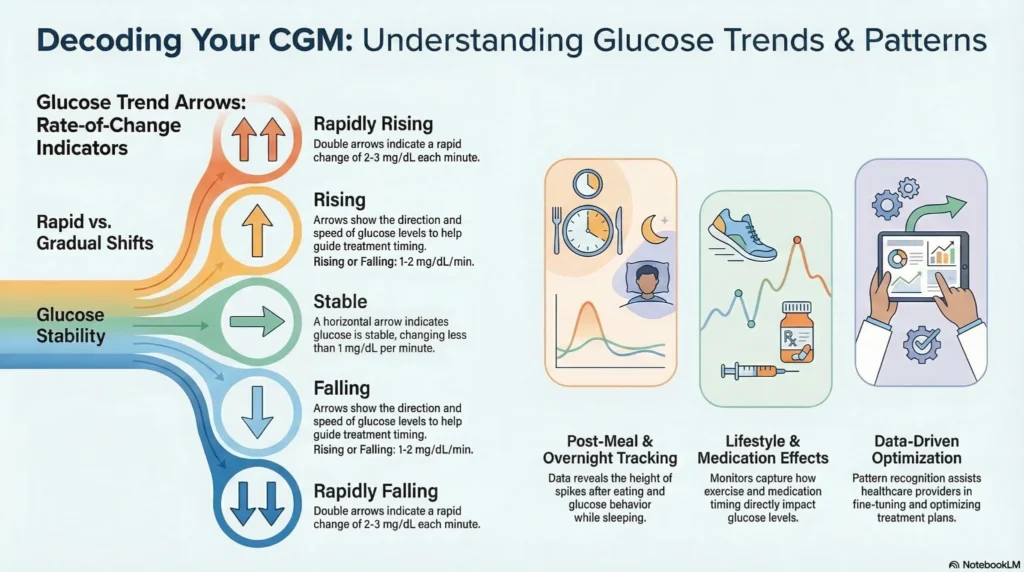
CGM and flash monitors display trend arrows indicating glucose direction:
| Arrow | Meaning | Rate of Change |
| ↑↑ | Rising rapidly | >2-3 mg/dL/min |
| ↑ | Rising | 1-2 mg/dL/min |
| → | Stable | <1 mg/dL/min |
| ↓ | Falling | 1-2 mg/dL/min |
| ↓↓ | Falling rapidly | >2-3 mg/dL/min |
Trend information helps anticipate glucose changes and guide treatment timing.
Pattern Recognition
Continuous data reveals glucose patterns:
- Post-meal spikes and duration
- Overnight glucose trends
- Exercise-related glucose changes
- Medication timing effects
Pattern recognition assists healthcare providers in treatment optimization.
Alert Customization
Programmable Alert Thresholds
CGM systems allow customization of:
- Low glucose alerts: Typically 70-80 mg/dL
- High glucose alerts: Individualized based on targets
- Urgent low alerts: 55 mg/dL (non-adjustable on some systems)
- Predictive alerts: Warn before reaching threshold
Alert Fatigue Management
- Excessive alerts can lead to alert fatigue where users ignore notifications:
- Review alert frequency with healthcare providers
- Set thresholds appropriate to individual glucose control
- Use temporary alert silencing during stable periods
- Adjust alert volumes and vibration settings
Understanding Glucose Metrics
Time-in-Range
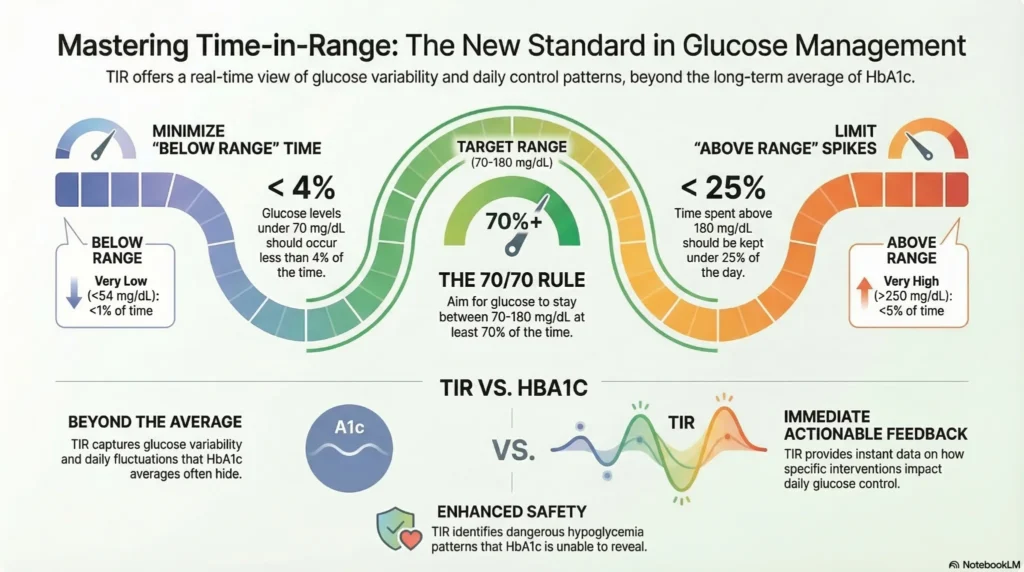
Definition and Clinical Targets
Time-in-Range (TIR) represents percentage of time glucose stays within target:
| Range | Target (mg/dL) | Goal |
| Target Range | 70-180 | >70% of time |
| Below Range | <70 | <4% of time |
| Very Low | <54 | <1% of time |
| Above Range | >180 | <25% of time |
| Very High | >250 | <5% of time |
Goals may vary based on individual factors including age, pregnancy, and hypoglycemia awareness.[11]
TIR vs. HbA1c
TIR provides additional information beyond HbA1c:
- Shows glucose variability (not just average)
- Reflects daily glucose control patterns
- May identify hypoglycemia HbA1c doesn’t reveal
- Provides immediate feedback on interventions
Both metrics complement each other in diabetes management.
Glucose Variability
Coefficient of Variation
Glucose variability measured as:
CV = (Standard Deviation ÷ Mean Glucose) × 100
- CV <36%: Target for stable glucose control
- CV >36%: Indicates high glucose variability
High variability may increase diabetes complication risk independent of average glucose.
Managing Variability
- Factors contributing to glucose variability:
- Sleep quality and stress
- Inconsistent carbohydrate intake
- Irregular meal timing
- Variable physical activity
- Medication adherence
Ambulatory Glucose Profile
Standardized Report Format
The Ambulatory Glucose Profile (AGP) presents CGM data in clinical format:
AGP Components:
- Median glucose curve (typical daily pattern)
- Interquartile range (25th-75th percentile)
- 10th-90th percentile range
- Target range visualization
- Daily glucose profiles overlaid
Clinical Interpretation
AGP reports help healthcare providers:
- Identify consistent patterns vs. one-time events
- Assess overnight glucose control
- Evaluate post-meal glucose responses
- Guide medication timing adjustments
Patients can generate AGP reports for medical appointments through device software.
Frequently Asked Questions About Glucose Monitors
How painful are CGM sensors when inserted?
Most users report feeling a quick pinch or no pain at all during insertion. The sensor needle is thinner than most finger-prick lancets and automates insertion in less than one second. Many users describe the sensation as similar to removing an adhesive bandage quickly. Children as young as 2 years old wear CGM sensors comfortably. After insertion, the majority of users report no ongoing discomfort during the 7-14 day wear period.
Can I shower, swim, or exercise with a CGM?
Yes. Modern CGM sensors are water-resistant and designed for daily life. Most systems are rated for:
Showering and bathing without restrictions
Swimming (typically up to 8 feet depth for 24 hours—check specific device ratings)
High-intensity exercise including contact sports (with proper adhesive overlay if needed)
Hot tubs and saunas (check manufacturer guidelines for temperature limits)
The sensor adhesive is designed to withstand sweat and water exposure. Many users apply additional adhesive overlays (sold separately) for extra security during intense activities or extended water exposure.
How long does insurance approval take for a CGM?
Timeline varies by insurance carrier and individual circumstances:
Pre-authorization review: 3-10 business days typically
Approval notification: 1-3 days after decision
Device shipment: 3-7 days after approval
Total timeline: Usually 1-3 weeks from initial submission to receiving the device
If initially denied: 78% of denials are overturned on appeal. The appeals process adds 2-4 weeks but often succeeds when proper medical documentation supports diabetes management need.
What if my CGM sensor fails or gives inaccurate readings?
Sensor failures are relatively rare but do occur:
Failure rate: Less than 2% of users experience sensor failure per month
Manufacturer replacement: All major manufacturers replace faulty sensors free of charge when you contact their technical support
Backup plan: Always keep a traditional blood glucose meter and test strips as backup for CGM malfunctions or when making treatment decisions during suspected inaccuracy
Warning period: Most systems alert you 12-24 hours before sensor expiration, allowing time to prepare replacement
Do I need a smartphone to use a CGM?
Not always. Options include:
Smartphone-based systems: Dexcom G7, FreeStyle Libre 3 (require compatible smartphone)
Dedicated receiver options: Some manufacturers offer standalone receivers (separate purchase) for those without smartphones or preferring not to use phones
Hybrid approach: Many users carry both phone and receiver for redundancy
Check compatibility lists on manufacturer websites—not all smartphones work with all CGM systems. iPhone and Android compatibility varies by model year.
How accurate are CGMs compared to fingerstick meters?
Modern CGMs approach fingerstick meter accuracy:
CGM accuracy (MARD): 8-10% for leading systems (Dexcom G7: 8.1%, FreeStyle Libre 3: 9.2%)
Fingerstick meter requirement: ±15% at glucose ≥100 mg/dL
Clinical significance: Both are sufficiently accurate for treatment decisions when used correctly
Key difference: CGMs measure interstitial fluid glucose, which lags blood glucose by 5-10 minutes during rapid changes. During stable periods, readings align closely.
Will my insurance cover a CGM?
Coverage depends on multiple factors:
Medicare: Covers CGMs for insulin users meeting specific criteria (effective 2023 expansion)
Private insurance: Most major insurers cover CGMs for Type 1 diabetes; Type 2 coverage varies and typically requires intensive insulin therapy
Medicaid: Coverage varies significantly by state
Documentation required: Usually needs prescription, diabetes diagnosis documentation, and evidence of insulin use or specific medical need
If not covered: Patient assistance programs from manufacturers (Dexcom, Abbott, Medtronic) may reduce costs to $0-75/month for qualifying individuals. See Costs section for details.
How often do I need to replace CGM sensors?
Replacement schedule depends on system:
Dexcom G7: Every 10 days
FreeStyle Libre 3: Every 14 days
Medtronic Guardian: Every 7 days
Eversense: Every 90-180 days (implantable, requires minor procedure for replacement)
Wearing sensors beyond manufacturer-approved duration may compromise accuracy and is not recommended.
Can I use a CGM if I’m pregnant or planning pregnancy?
Yes, with medical guidance. CGMs are increasingly used during pregnancy for tighter glucose control:
Gestational diabetes: Some CGM systems have FDA approval for pregnancy
Pre-existing diabetes: CGMs help achieve stricter glucose targets needed during pregnancy
Medical supervision required: More frequent healthcare provider communication needed to adjust targets and interpret data
Always discuss CGM use with your obstetrician and endocrinologist before and during pregnancy.
Data Management and Sharing
Cloud Platforms and Apps
Manufacturer Platforms
Each CGM system provides data management:
- Dexcom Clarity: Web and mobile app with AGP reports
- LibreView: FreeStyle Libre data platform
- CareLink: Medtronic data management
Features typically include:
- Glucose graphs and statistics
- Downloadable reports for healthcare providers
- Pattern recognition insights
- Meal and activity logs (if entered)
Telehealth Integration
- Remote data review by healthcare providers
- Pre-visit report generation
- Asynchronous communication about patterns
- Reduced need for in-person appointments for stable patients
Data Security Considerations
User Responsibilities:
- Strong passwords for app accounts
- Two-factor authentication (when available)
- Regular app updates for security patches
- Review of connected devices and permissions
Platform Security:
- Privacy policy review recommended
- HIPAA compliance (for medical platforms)
- Encrypted data transmission
- Secure cloud storage
Connect with the Glucose Monitoring Community
You’re not alone in navigating diabetes technology. These resources provide support, real-world experiences, and ongoing education:
Online Communities and Support Groups
- Diabetes Daily Forums (diabetesdaily.com): Active community discussing all device types with user reviews
- Beyond Type 1 (beyondtype1.org): Type 1-focused organization with CGM troubleshooting guides
- TuDiabetes (tudiabetes.org): Peer support community with device comparison discussions
- Facebook Groups: “CGM Users,” “FreeStyle Libre Users,” “Dexcom G7 Community” (search Facebook for closed support groups)
Video Resources
- Search YouTube for device-specific channels: “Dexcom sensor insertion,” “FreeStyle Libre application,” “CGM accuracy testing”
- Diabetes educators often post troubleshooting tips and comparison videos
- User testimonials provide realistic expectations beyond manufacturer marketing
Manufacturer Resources
- 24/7 Technical Support Hotlines: All major manufacturers provide phone support for device issues
- Educational Webinars: Free training sessions on maximizing device benefits
- Customer Portals: Access to replacement supplies, insurance assistance, and troubleshooting guides
Healthcare Provider Collaboration
- Certified Diabetes Educators (CDEs): Specialists who provide device training and data interpretation
- Endocrinology Offices: Many have manufacturer representatives visit regularly with sample devices and application demonstrations
Share This Resource
Found this guide helpful? Share with others navigating glucose monitoring decisions:
- – Send to family members helping with diabetes management
- – Share in diabetes support groups or forums
- – Print for discussion at your next doctor appointment
- – Forward to friends newly diagnosed with diabetes
When to Consult a Healthcare Provider
Glucose monitoring device selection and use should involve healthcare provider guidance. Specific situations requiring medical consultation include:
- Initiating any glucose monitoring for diabetes management
- Changing from traditional meters to CGM or flash monitors
- Interpreting glucose patterns for treatment adjustments
- Experiencing frequent or severe hypoglycemia
- Considering insulin pump integration
- Pregnancy with diabetes or gestational diabetes
- CGM readings not correlating with symptoms
- Sensor accuracy concerns or frequent errors
- Determining appropriate monitoring frequency
- Insurance coverage questions or prior authorization needs
This guide provides educational information only and does not replace individualized medical advice. All monitoring approaches and device selections should be discussed with your healthcare team.
References
[1] Juvenile Diabetes Research Foundation Continuous Glucose Monitoring Study Group. (2008). Continuous glucose monitoring and intensive treatment of type 1 diabetes. New England Journal of Medicine, 359(14), 1464-1476.
[2] Beck, R. W., et al. (2017). Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: The DIAMOND randomized clinical trial. JAMA, 317(4), 371-378.
[3] Martens, T., et al. (2021). Effect of continuous glucose monitoring on glycemic control in patients with type 2 diabetes treated with basal insulin: A randomized clinical trial. JAMA, 325(22), 2262-2272.
[4] Bolinder, J., et al. (2016). Novel glucose-sensing technology and hypoglycaemia in type 1 diabetes: A multicentre, non-masked, randomised controlled trial. Lancet, 388(10057), 2254-2263.
[5] U.S. Food and Drug Administration. (2020). Self-Monitoring Blood Glucose Test Systems for Over-the-Counter Use: Guidance for Industry and Food and Drug Administration Staff.
[6] Klonoff, D. C., et al. (2022). The pursuit of noninvasive glucose monitoring: A review of currently available devices and recent advances. Diabetes Technology & Therapeutics, 24(11), 739-752.
[7] Wadwa, R. P., et al. (2022). Accuracy of a factory-calibrated, real-time continuous glucose monitoring system during 10 days of use in youth and adults with diabetes. Diabetes Technology & Therapeutics, 24(2), 95-105.
[8] Castorino, K., et al. (2022). Performance evaluation of the FreeStyle Libre 3 system: Real-world evidence from a prospective observational study. Diabetes Technology & Therapeutics, 24(8), 540-548.
[9] Centers for Medicare & Medicaid Services. (2023). National Coverage Determination for Continuous Glucose Monitors (CGMs) (CAG-00459N).
[10] U.S. Food and Drug Administration. (2022). General Wellness: Policy for Low Risk Devices – Guidance for Industry and Food and Drug Administration Staff.
[11] American Diabetes Association. (2024). Glycemic targets: Standards of care in diabetes—2024. Diabetes Care, 47(Supplement_1), S111-S125.
[12] Aleppo, G., et al. (2022). MOBILE study: Performance and patient satisfaction with the MiniMed 780G system. Diabetes Technology & Therapeutics, 24(9), 607-615.
[13] Reddy, M., et al. (2023). Effect of continuous glucose monitoring on quality of life and glucose control in type 2 diabetes: A meta-analysis. Diabetes Care, 46(5), 1033-1041.
[14] American Diabetes Association. (2024). Classification and diagnosis of diabetes: Standards of care in diabetes—2024. Diabetes Care, 47(Supplement_1), S20-S42.
