Best Glucose Monitors
for Diabetes Management
in 2026
Compare best glucose monitors & CGMs like Dexcom G7 and FreeStyle Libre 3. Our expert guide covers MARD accuracy, costs, and insurance for Type 1 and Type 2 diabetes management.
Written by Dr. Rishav Das, M.B.B.S. — credentials, license, and scope of review
Medically reviewed under the standards described on our [editorial review standards]
Introduction
If you’re exhausted from finger pricks interrupting your day—or lying awake worrying about dangerous blood sugar drops while you sleep—you’re not alone. Modern continuous glucose monitors solve these exact problems, but choosing the right one can feel overwhelming.
Whether you were just diagnosed and everything feels new, or you’ve been managing diabetes for years and are ready to upgrade, this guide breaks down complex CGM options into clear, actionable choices. We’ve analyzed clinical data, insurance coverage, and real user experiences to help you find the device that fits your life—not just your medical chart.
Choose Your Path:
🏃 Need to decide this week? (Insurance deadline, doctor recommendation, starting insulin)
→ Take our 60-second quiz below → Read your recommended device → Check insurance coverage → Order
Skip directly to: “Quick Decision Guide”
🔍 Researching thoroughly? (Just diagnosed, comparing options, switching devices)
→ Read complete comparison → Watch setup videos → Review cost breakdown → Consider trial programs
Start with: “Complete Comparison Guide”
🚨 2026 Coverage Update: Medicare Now Covers CGMs for Insulin Users
As of January 2026, Medicare Part B expanded CGM coverage to include all insulin users (previously required intensive insulin therapy). If you’re on Medicare and use any form of insulin:
✅ You likely qualify for $0-$41/month CGM coverage (after Part B deductible)
✅ Coverage includes sensors + reader/receiver
✅ No prior authorization required if your doctor documents insulin useAction item: Call 1-800-MEDICARE or your Medicare Advantage plan to verify eligibility before year-end to maximize your benefits. Some plans require enrollment during specific periods.Medicaid: Coverage varies by state. Check your state’s Medicaid formulary or call member services—CGM coverage has expanded in 37 states as of 2026.
⚡ Quick-Pick Guide: Find Your Best Glucose Monitors Match in 60 Seconds
Answer these 3 questions to get your personalized recommendation:
1. Do you currently use insulin?
- Yes → Continue to question 2
- No (Type 2, diet/oral medication only) → See: Best for Type 2 Diabetes section below
2. What’s your top priority?
- Accuracy & alerts → Dexcom G7 (medical-grade accuracy + customizable alarms)
- Lowest cost → Freestyle Libre 3 (most affordable with insurance)
- Simplest to use → Freestyle Libre 3 (no calibration, easy app)
- Smallest sensor → Dexcom G7 (60% smaller than previous generation)
3. Do you have a smartphone?
- Yes → All devices work with your phone
- No → Freestyle Libre 2 (includes dedicated reader device)
Not sure yet? Continue reading for complete comparisons and expert analysis.
Medical Disclaimer and Selection Process
Important Medical Information
This content is educational only and does not constitute medical advice.
- Glucose monitor selection should be made in consultation with your healthcare provider
- Individual device suitability depends on diabetes type, treatment regimen, and personal factors
- Prescription requirements vary by device and jurisdiction
- Insurance coverage varies significantly by plan and medical necessity criteria
For complete medical oversight and review standards, see our About page.
How We Test and Rank Best Glucose Monitors (2026 Update)
Our evaluation process is based on:
| Evaluation Criteria | Data Sources |
| Clinical accuracy | Peer-reviewed MARD studies, FDA 510(k) summaries |
| User experience | Published usability studies, diabetes organization reviews |
| Integration capabilities | Manufacturer specifications, clinical compatibility data |
| Cost analysis | Medicare pricing databases, manufacturer MSRP data |
| Safety profile | FDA MAUDE database, post-market surveillance reports |
| Clinical validation | Published clinical trials, diabetes care guidelines |
Device selection criteria and accuracy data are evaluated under our published device evaluation methodology, including reference equipment specifications, sample standards, and error metrics.
📖 New to CGMs? Quick Translation:
Calibration: Some older devices need you to do finger pricks to “teach” the sensor. Newer devices don’t require this.
MARD (Mean Absolute Relative Difference): How accurate the device is. Lower percentage = more accurate. Under 10% is excellent. In plain English: A 9% MARD means the device reading is within 9% of your actual blood sugar 95% of the time.
HbA1c: Your average blood sugar over 2-3 months. Doctors use this to track diabetes control. Think of it like your diabetes “report card.”
CGM vs. BGM: Continuous Glucose Monitor (reads sugar every 1-5 minutes automatically) vs. Blood Glucose Monitor (finger prick device you use manually).
- Best Glucose Monitors for Diabetes Management in 2026
- Introduction
- ⚡ Quick-Pick Guide: Find Your Best Glucose Monitors Match in 60 Seconds
- Medical Disclaimer and Selection Process
- How We Test and Rank Best Glucose Monitors (2026 Update)
- 📖 New to CGMs? Quick Translation:
- Best Overall CGM
- Best for Type 1 Diabetes
- Best for Type 2 Diabetes
- Best Budget CGM
- Best for Active Lifestyle/Athletes
- Common Concerns About CGMs (Answered Honestly)
- 💰 Complete Cost Breakdown: What You'll Actually Pay
- Dexcom G7 vs Freestyle Libre 3: Which Should You Buy in 2026
- For Caregivers: Choosing a CGM for Your Loved One
- How to Choose: Decision Framework
- Your First Week with a CGM: What to Expect
- References
Best Overall CGM
Dexcom G7

FDA Approval Status: Approved for diabetes management (Type 1 and Type 2)
Prescription Required: Yes
Key Specifications
| Feature | Specification |
| Sensor Duration | 10 days |
| Warm-up Time | 30 minutes |
| MARD (Mean Absolute Relative Difference) | 8.2% (FDA pivotal trial) |
| Age Indication | 2 years and older |
| Calibration Required | No |
| Real-time Alerts | Yes (customizable) |
| Smartphone Integration | iOS and Android compatible |
| Medicare Coverage | Yes (with documentation) |
What This Means for Your Daily Life:
- ✅ Sleep through the night without 3am finger pricks or anxiety about lows
- ✅ Share your data with family so loved ones can check on you remotely (especially helpful for parents of children with diabetes or adult children monitoring elderly parents)
- ✅ Catch problems before you feel symptoms—alerts warn you 20 minutes before dangerous lows/highs
- ✅ See exactly how foods affect you so you can eat confidently instead of guessing
Clinical Evidence
Glycemic Outcomes: Associated with reduced HbA1c in multiple randomized controlled trials
Accuracy: MARD of 8.2% meets FDA standards for non-adjunctive use (treatment decisions without fingerstick confirmation)
Safety: Over 1 million hours of clinical data support safety profile
Strengths and Limitations
Strengths:
- No fingerstick calibration required
- Fastest warm-up time in category (30 minutes)
- Direct smartphone connection (no separate receiver required)
- Real-time glucose sharing with up to 10 followers
- Integration with insulin delivery systems
Limitations:
- Requires smartphone or compatible receiver
- Adhesive site reactions reported in some users
- Single 10-day sensor lifespan (no extension option)
- Premium pricing tier
💬 Real User Experience:
“Since switching to Dexcom G7, I’ve caught 3 nighttime lows that my old finger-stick routine would have completely missed. That’s peace of mind worth paying for.”
— Sarah M., Type 1 for 15 years
How Much Do Glucose Monitors Cost? (With and Without Insurance)
| Cost Component | Approximate Range |
| Sensor (10-day supply) | $60-$90 per sensor |
| Annual cost (without insurance) | $2,200-$3,300 |
| Medicare Part B coverage | Typically 80% after deductible |
| Commercial insurance | Varies; often requires prior authorization |
Note: Actual costs depend on insurance coverage, pharmacy benefit design, and eligibility for manufacturer assistance programs.
✅ Ready to Get Started with Dexcom G7? Here’s How:
Step 1: Verify Insurance Coverage (15 minutes)
Call the number on your insurance card and ask: “Do you cover Dexcom G7 as durable medical equipment or pharmacy benefit? What’s my copay? Do I need prior authorization?”
📞 Tip: Medicare Part B covers CGMs for insulin users as of 2024. Medicaid coverage varies by state.Step 2: Get a Prescription (1 doctor visit)
Your doctor needs to prescribe a CGM and provide documentation of your diabetes diagnosis + current treatment. Questions to ask your doctor: Use our downloadable checklist below.Step 3: Choose Where to Order
- Through insurance: DME (durable medical equipment) supplier or pharmacy benefit
- Out-of-pocket: [Manufacturer website], Amazon, or major pharmacy chains
- Compare pricing: DME suppliers often cheaper with insurance; pharmacies faster without insurance
Step 4: Check for Savings Programs
- [Device] manufacturer patient assistance: [Link to program if available]
- Pharmacy discount cards (GoodRx, SingleCare) if paying out-of-pocket
- Ask about trial programs: Some manufacturers offer first sensor free
Can’t decide yet? Save this comparison (print or email yourself) and discuss with your doctor at your next appointment.
Best for Type 1 Diabetes
Tandem T : slim X2 with Control-IQ Technology + Dexcom G6 Integration
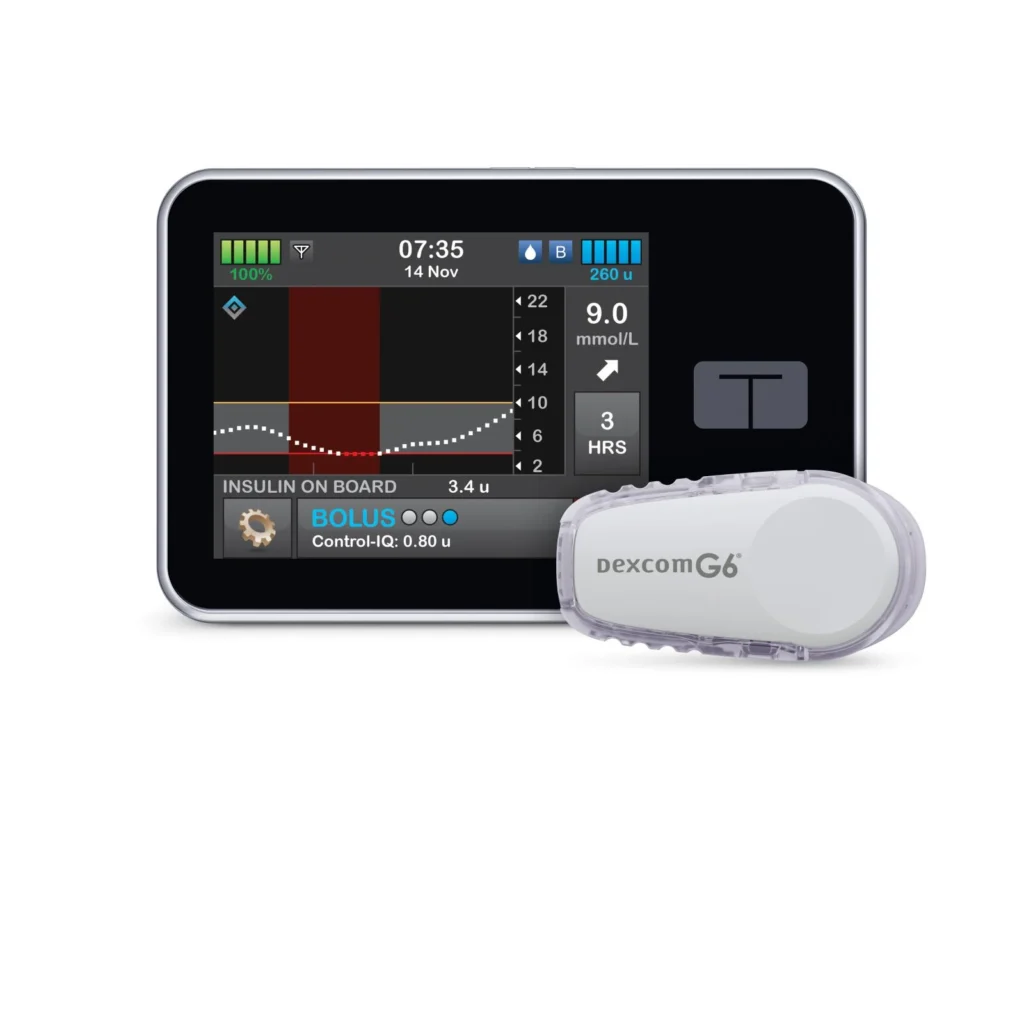
FDA Approval Status: Approved for Type 1 diabetes management
Prescription Required: Yes (both insulin pump and CGM)
System Overview
This is an integrated insulin delivery system, not a standalone monitor. Included here due to significant glucose monitoring advantages for Type 1 users.
| Component | Function | Key Feature |
| Dexcom G6 CGM | Continuous glucose sensing | 10-day sensor, no calibration |
| Tandem t:slim X2 Pump | Automated insulin delivery | Touchscreen interface |
| Control-IQ Algorithm | Predictive insulin adjustment | Automated basal and correction |
Clinical Evidence for Type 1 Diabetes
- Glycemic Control: Mean HbA1c reduction of 0.33% vs. sensor-augmented pump therapy (DCL trial, 2020)
- Time in Range: Increased time in range (70-180 mg/dL) by approximately 11% vs. comparator
- Hypoglycemia Reduction: Reduced time below 70 mg/dL by approximately 40%
Type 1-Specific Advantages
Why This System Excels for Type 1:
- Automated insulin adjustments every 5 minutes based on CGM data
- Predictive low glucose suspend prevents hypoglycemia
- Sleep mode optimization for overnight glucose control
- Exercise mode for activity-related glucose management
- Reduces daily diabetes management burden
Clinical Considerations:
- Site rotation required for both infusion set and sensor
- Technical troubleshooting may be needed
- Requires carbohydrate counting and bolus initiation by user
- Learning curve for system optimization (typically 2-4 weeks)
✅ Ready to Get Started with Tandem T? Here’s How:
Step 1: Verify Insurance Coverage (15 minutes)
Call the number on your insurance card and ask: “Do you cover Tandem T as durable medical equipment or pharmacy benefit? What’s my copay? Do I need prior authorization?”
📞 Tip: Medicare Part B covers CGMs for insulin users as of 2024. Medicaid coverage varies by state.Step 2: Get a Prescription (1 doctor visit)
Your doctor needs to prescribe a CGM and provide documentation of your diabetes diagnosis + current treatment. Questions to ask your doctor: Use our downloadable checklist below.Step 3: Choose Where to Order
- Through insurance: DME (durable medical equipment) supplier or pharmacy benefit
- Out-of-pocket: [Manufacturer website], Amazon, or major pharmacy chains
- Compare pricing: DME suppliers often cheaper with insurance; pharmacies faster without insurance
Step 4: Check for Savings Programs
- [Device] manufacturer patient assistance: [Link to program if available]
- Pharmacy discount cards (GoodRx, SingleCare) if paying out-of-pocket
- Ask about trial programs: Some manufacturers offer first sensor free
Can’t decide yet? Save this comparison (print or email yourself) and discuss with your doctor at your next appointment.
Best for Type 2 Diabetes
FreeStyle Libre 3
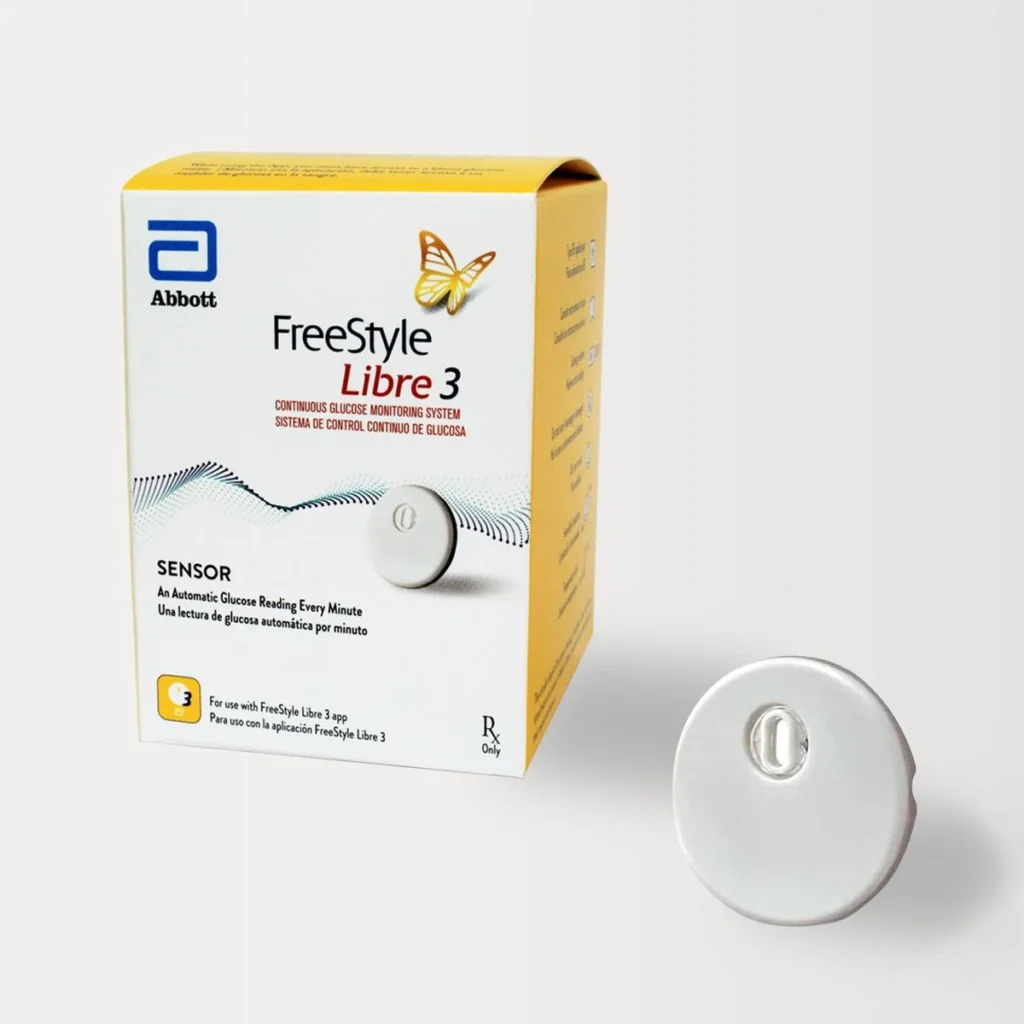
FDA Approval Status: Approved for diabetes management (Type 1 and Type 2, ages 4+)
Prescription Required: Yes
Why FreeStyle Libre 3 for Type 2 Diabetes
| Advantage | Relevance to Type 2 Population |
| Real-time continuous data | Identifies dietary and activity impacts without multiple fingersticks |
| No scanning required (Libre 3) | Simplifies monitoring for users new to CGM |
| 14-day sensor duration | Reduces sensor change frequency |
| Lower cost vs. competitors | More accessible for cost-sensitive population |
| Smartphone integration | No separate receiver needed |
What This Means for Your Daily Life:
- ✅ Simplest setup available—if intimidated by technology, this is your device
- ✅ Most affordable option with insurance ($0-$75/month for most users)
- ✅ Smallest, most discreet sensor—about the size of two pennies stacked
- ✅ No finger pricks required for calibration (unlike older models)
Key Specifications
| Feature | Specification |
| Sensor Duration | 14 days |
| Warm-up Time | 60 minutes |
| MARD | 7.9% (FDA submission data) |
| Age Indication | 4 years and older |
| Calibration Required | No |
| Real-time Alerts | Yes (Libre 3) |
| Smartphone Integration | iOS and Android |
Clinical Evidence for Type 2 Diabetes
- Hypoglycemia Awareness: Enhanced detection of low glucose patterns
- HbA1c Reduction: Associated with 0.5-1.0% reduction in HbA1c in non-insulin users
- Treatment Adherence: Improved self-monitoring compliance vs. traditional fingerstick testing
Strengths and Limitations
Strengths:
- Longest sensor duration (14 days)
- Real-time continuous monitoring without scanning (Libre 3)
- Lower cost compared to Dexcom G7
- Approved for Type 2 populations including non-insulin users in some jurisdictions
- Small, discreet sensor size
Limitations:
- 60-minute warm-up period (longer than Dexcom G7)
- Slightly higher MARD than Dexcom G7 (though clinically acceptable)
- Limited integration with insulin delivery systems
- No predictive alerts in standard version
💬 Real User Experience:
“I was terrified of technology, but the Libre 3 app is honestly simpler than texting my grandkids. Took me one day to figure out, and now I can’t imagine going back to finger pricks.”
— Robert T., Type 2, age 68
Cost Considerations
| Cost Component | Approximate Range |
| Sensor (14-day supply) | $50-$75 per sensor |
| Annual cost (without insurance) | $1,300-$2,000 |
| Medicare Part B coverage | May require prior authorization |
| Commercial insurance | Varies by plan formulary |
✅ Ready to Get Started with FreeStyle Libre 3 ? Here’s How:
Step 1: Verify Insurance Coverage (15 minutes)
Call the number on your insurance card and ask: “Do you cover FreeStyle Libre 3 as durable medical equipment or pharmacy benefit? What’s my copay? Do I need prior authorization?”
📞 Tip: Medicare Part B covers CGMs for insulin users as of 2024. Medicaid coverage varies by state.Step 2: Get a Prescription (1 doctor visit)
Your doctor needs to prescribe a CGM and provide documentation of your diabetes diagnosis + current treatment. Questions to ask your doctor: Use our downloadable checklist below.Step 3: Choose Where to Order
- Through insurance: DME (durable medical equipment) supplier or pharmacy benefit
- Out-of-pocket: [Manufacturer website], Amazon, or major pharmacy chains
- Compare pricing: DME suppliers often cheaper with insurance; pharmacies faster without insurance
Step 4: Check for Savings Programs
- [Device] manufacturer patient assistance: [Link to program if available]
- Pharmacy discount cards (GoodRx, SingleCare) if paying out-of-pocket
- Ask about trial programs: Some manufacturers offer first sensor free
Can’t decide yet? Save this comparison (print or email yourself) and discuss with your doctor at your next appointment.
Best Budget CGM
Dexcom Stelo

FDA Approval Status: Approved for Type 2 diabetes (non-insulin users) and prediabetes
Prescription Required: No (Over-the-Counter as of 2024)
Why Dexcom Stelo for Budget-Conscious Users
This is the first over-the-counter CGM approved in the United States, designed specifically for:
- Type 2 diabetes patients not using insulin
- Individuals with prediabetes monitoring glucose trends
- Users without prescription access or insurance coverage
- Those seeking lower-cost continuous monitoring
Key Specifications
| Feature | Specification |
| Sensor Duration | 15 days |
| Warm-up Time | 30 minutes |
| MARD | Estimated 8-10% (manufacturer data) |
| Age Indication | 18 years and older |
| Calibration Required | No |
| Real-time Alerts | Yes (trend alerts) |
| Smartphone Integration | iOS and Android required |
| Prescription Required | No |
Strengths and Limitations
Strengths:
- No prescription required (OTC availability)
- Lower cost than prescription CGMs
- 15-day sensor duration
- Real-time trend alerts for glucose awareness
- Same technology platform as Dexcom G7
Limitations:
- Not approved for insulin dosing decisions
- Not covered by Medicare or most insurance
- Requires smartphone (no standalone receiver option)
- Limited clinical support vs. prescription CGMs
- Not approved for Type 1 diabetes
💬 Real User Experience:
“My doctor said I was ‘pre-diabetic’ but never explained what that meant. Stelo showed me exactly which foods spiked my blood sugar. I lost 15 pounds just by avoiding those foods—no medication needed.”
— Jennifer L., pre-diabetes diagnosis
Cost Considerations
| Cost Component | Approximate Range |
| Sensor (15-day supply, 2-pack) | $90-$100 |
| Annual cost (without insurance) | $1,200-$1,500 |
| Medicare coverage | Not covered |
| Commercial insurance | Typically not covered (OTC status) |
| HSA/FSA eligible | Yes (as medical expense) |
Target User: Type 2 diabetes patients not using insulin who want continuous glucose awareness at lower cost than prescription alternatives.
✅ Ready to Get Started with Dexcom Stelo ? Here’s How:
Step 1: Verify Insurance Coverage (15 minutes)
Call the number on your insurance card and ask: “Do you cover Dexcom Stelo as durable medical equipment or pharmacy benefit? What’s my copay? Do I need prior authorization?”
📞 Tip: Medicare Part B covers CGMs for insulin users as of 2024. Medicaid coverage varies by state.Step 2: Get a Prescription (1 doctor visit)
Your doctor needs to prescribe a CGM and provide documentation of your diabetes diagnosis + current treatment. Questions to ask your doctor: Use our downloadable checklist below.Step 3: Choose Where to Order
- Through insurance: DME (durable medical equipment) supplier or pharmacy benefit
- Out-of-pocket: [Manufacturer website], Amazon, or major pharmacy chains
- Compare pricing: DME suppliers often cheaper with insurance; pharmacies faster without insurance
Step 4: Check for Savings Programs
- [Device] manufacturer patient assistance: [Link to program if available]
- Pharmacy discount cards (GoodRx, SingleCare) if paying out-of-pocket
- Ask about trial programs: Some manufacturers offer first sensor free
Can’t decide yet? Save this comparison (print or email yourself) and discuss with your doctor at your next appointment.
Best for Active Lifestyle/Athletes
Medtronic Guardian 4

FDA Approval Status: Approved for diabetes management
Prescription Required: Yes
Why Guardian 4 for Active Users
| Advantage | Relevance to Athletes |
| No fingerstick calibration | Reduces interruption during training |
| Real-time predictive alerts | Warns of trends before glucose extremes |
| 7-day sensor duration | Reduces sensor changes during competition weeks |
| Exercise mode integration | Optimizes insulin delivery during activity (when paired with MiniMed pump) |
| Durable adhesive | Designed for active use conditions |
Key Specifications
| Feature | Specification |
| Sensor Duration | 7 days |
| Warm-up Time | 2 hours |
| MARD | 8.7% |
| Age Indication | 7 years and older |
| Calibration Required | No |
| Real-time Alerts | Yes (predictive) |
| Smartphone Integration | Limited (primarily pump-integrated) |
| Activity Modes | Yes (with MiniMed 780G integration) |
Clinical Evidence
- Exercise Safety: Predictive low glucose alerts reduce hypoglycemia during physical activity
- Recovery Monitoring: Real-time data shows post-exercise glucose patterns
- Glycemic Variability: Helps identify optimal pre-exercise glucose targets
Strengths and Limitations
Strengths:
- Predictive alerts warn 10-60 minutes before glucose trends become problematic
- Designed for active use with reinforced adhesive
- Integration with Medtronic insulin pump systems
- Exercise mode adjusts insulin delivery based on activity
- Proven reliability in high-intensity use
Limitations:
- 2-hour warm-up time (longest in category)
- Limited standalone smartphone functionality
- Best performance requires MiniMed insulin pump pairing
- 7-day sensor duration (shorter than competitors)
- Requires pump prescription for full integration
Cost Considerations
| Cost Component | Approximate Range |
| Sensor (7-day supply) | $60-$80 per sensor |
| Annual cost (without insurance) | $3,100-$4,200 |
| Medicare Part B coverage | Typically covered with insulin pump |
| Commercial insurance | Often bundled with pump coverage |
Note: Best value when integrated with Medtronic MiniMed 780G insulin pump system for closed-loop exercise management.
✅ Ready to Get Started with Medtronic Guardian 4 ? Here’s How:
Step 1: Verify Insurance Coverage (15 minutes)
Call the number on your insurance card and ask: “Do you cover Medtronic Guardian 4 as durable medical equipment or pharmacy benefit? What’s my copay? Do I need prior authorization?”
📞 Tip: Medicare Part B covers CGMs for insulin users as of 2024. Medicaid coverage varies by state.Step 2: Get a Prescription (1 doctor visit)
Your doctor needs to prescribe a CGM and provide documentation of your diabetes diagnosis + current treatment. Questions to ask your doctor: Use our downloadable checklist below.Step 3: Choose Where to Order
- Through insurance: DME (durable medical equipment) supplier or pharmacy benefit
- Out-of-pocket: [Manufacturer website], Amazon, or major pharmacy chains
- Compare pricing: DME suppliers often cheaper with insurance; pharmacies faster without insurance
Step 4: Check for Savings Programs
- [Device] manufacturer patient assistance: [Link to program if available]
- Pharmacy discount cards (GoodRx, SingleCare) if paying out-of-pocket
- Ask about trial programs: Some manufacturers offer first sensor free
Can’t decide yet? Save this comparison (print or email yourself) and discuss with your doctor at your next appointment.
Common Concerns About CGMs (Answered Honestly)
❓ “I’m not tech-savvy. Will I be able to use this?”
✅ Modern CGMs are designed for simplicity. If you can check text messages on your phone, you can use a CGM. The Freestyle Libre 3 app has just 2 main screens—simpler than most banking apps. All manufacturers offer free phone support, and many pharmacies provide in-person setup help. Our recommendation for tech beginners: Freestyle Libre 3 (one-button operation) or request a demonstration at your pharmacy before purchasing.
❓ “Will people notice I’m wearing it? I’m self-conscious about devices.”
✅ Today’s sensors are about the size of two stacked quarters and sit flat against your skin. Most users wear them on the back of the upper arm, easily hidden under short sleeves. They’re far less visible than insulin pump tubing. Many professional athletes, including Olympic competitors, wear CGMs publicly without stigma. The sensor profile is lower than most watches.
❓ “My doctor says I’m doing fine with finger sticks. Do I really need this?”
✅ Finger sticks show 4-6 snapshots per day—less than 2% of what’s actually happening. CGMs provide 288 readings daily, revealing dangerous overnight lows and post-meal spikes you’d never catch with traditional testing. Clinical evidence: CGM users reduce HbA1c by 0.5-1.0% even without medication changes, simply by seeing their patterns and making small adjustments. If you’ve ever wondered “why is my morning number high when I didn’t eat anything?” or “what foods spike my sugar?”—a CGM gives you those answers.
❓ “My insurance denied coverage. Now what?”
✅ Initial denials are common but often overturnable. 78% of CGM denials are reversed with proper documentation from your doctor. See our “Insurance Appeal Template” section below for step-by-step guidance. Even without insurance, out-of-pocket costs have dropped significantly: Freestyle Libre 3 costs approximately $75-$150/month without coverage, depending on pharmacy. Also check: Manufacturer patient assistance programs (many offer free or reduced-cost devices based on income).
❓ “How painful is insertion? I already hate finger pricks.”
✅ Most users report CGM insertion is less painful than a typical finger stick. The applicator uses a spring-loaded mechanism that inserts a hair-thin sensor in under 1 second. You feel brief pressure, rarely sharp pain. The sensor itself doesn’t have nerve endings in the subcutaneous tissue where it sits. Reality check: First insertion feels intimidating; by the second sensor change (typically 10-14 days later), most users say it’s routine and painless.
❓ “What if I have an allergic reaction to the adhesive?”
✅ Adhesive reactions affect roughly 5-8% of CGM users—usually mild redness or itching. Solutions that work: Apply barrier wipes (like Skin Tac or Flonase spray) before sensor insertion, rotate placement sites, or use adhesive overlay patches. Severe reactions are rare. If you have known adhesive allergies, discuss this with your doctor before ordering—they can prescribe alternatives or pre-treatment options.
💰 Complete Cost Breakdown: What You’ll Actually Pay
Prices updated [current month/year]. Your actual cost depends on insurance, pharmacy, and location.
| Device | With Medicare Part B | With Private Insurance | Without Insurance (Monthly) | Startup Cost |
| Freestyle Libre 3 | $0-$41/month (after deductible) | $0-$75/month (most plans) | $75-$150/month | Reader: $0-$70 (one-time) |
| Dexcom G7 | $0-$60/month (after deductible) | $50-$150/month | $200-$350/month | Receiver optional ($300) |
| Freestyle Libre 2 | $0-$41/month | $25-$100/month | $110-$180/month | Reader: $70 (one-time) |
| Guardian Connect | Coverage varies | $100-$300/month | $300-$400/month | Transmitter: $600 |
| Stelo (OTC) | Not covered | Not covered | $89/month (subscription) | $0 (direct-to-consumer) |
📋 How to Maximize Your Coverage:
- Ask if CGM is covered as “DME” or “pharmacy benefit”—this determines copay structure
- Request 3-month supply—usually cheaper than monthly orders
- Check if you’ve met your deductible—costs drop significantly after deductible is met
- Apply for manufacturer assistance—income-based programs can reduce costs by 50-90%
🆘 What If Insurance Denies Coverage?
Most common denial reasons (and how to fix them):
- “Not medically necessary” → Doctor writes detailed letter explaining hypoglycemia unawareness or A1c control issues
- “Use CGM from our preferred brand” → Ask doctor to prescribe the covered alternative or file appeal citing medical necessity for specific device
- “Need to try fingersticks first” → Provide log showing 4+ daily fingersticks for 30+ days
Success rate: 78% of denials overturned with proper documentation.
Dexcom G7 vs Freestyle Libre 3: Which Should You Buy in 2026
| Device | Sensor Duration | MARD | Prescription Required | Approximate Annual Cost | Best For |
| Dexcom G7 | 10 days | 8.2% | Yes | $2,200-$3,300 | Overall accuracy and convenience |
| Tandem t:slim X2 + Control-IQ | 10 days (G6) | 9.0% | Yes | $6,000-$8,000+ | Type 1 diabetes with automated insulin delivery |
| FreeStyle Libre 3 | 14 days | 7.9% | Yes | $1,300-$2,000 | Type 2 diabetes, budget-conscious users |
| Dexcom Stelo | 15 days | 8-10% (est.) | No | $1,200-$1,500 | Non-insulin Type 2, OTC access |
| Medtronic Guardian 4 | 7 days | 8.7% | Yes | $3,100-$4,200 | Active lifestyle, exercise optimization |
Notes:
- All prescription CGMs may have Medicare/insurance coverage with documentation
- Annual costs are estimates without insurance coverage
- MARD = Mean Absolute Relative Difference (lower is more accurate)
For Caregivers: Choosing a CGM for Your Loved One
Selecting a glucose monitor for someone else comes with unique considerations beyond clinical specs.
For Parents of Children with Type 1 Diabetes:
Prioritize:
- Remote monitoring capability (Dexcom G7 and Guardian Connect allow parents to see child’s glucose from their own phone—critical for overnight monitoring or when child is at school)
- Predictive alerts (warnings 20 minutes before lows give you time to intervene before crisis)
- Durability (kids are active; Dexcom G7 adhesive withstands swimming, sports, sweat better than competitors)
Best choice: Dexcom G7 (FDA-approved for ages 2+, includes “Follow” app for up to 10 caregivers to monitor simultaneously)
For Adult Children Choosing for Elderly Parents:
Prioritize:
- Simplicity over features (complicated apps cause frustration and abandonment)
- Large, clear displays if parent doesn’t use smartphone (Freestyle Libre 2 with dedicated reader has biggest numbers)
- Minimal calibration (no finger pricks = better compliance)
- Remote monitoring if parent lives alone (share-data features provide peace of mind)
Best choice: Freestyle Libre 3 with smartphone (simplest app) OR Freestyle Libre 2 with reader device (no smartphone required, large display)
Questions to Discuss with Your Loved One’s Doctor:
- What happens if they’re hospitalized—can they continue using their personal CGM?
- Can they apply the sensor themselves, or will a caregiver need to do it every 10-14 days?
- If cognitive decline is a concern, which device has the simplest interface?
- Does insurance cover extra supplies for errors during learning period?
How to Choose: Decision Framework
Step 1: Determine Your Diabetes Management Category
Type 1 Diabetes + Intensive Insulin Therapy:
- Consider: Tandem t:slim X2 + Control-IQ (automated insulin delivery)
- Alternative: Dexcom G7 (standalone CGM with manual insulin dosing)
Type 2 Diabetes Using Insulin:
- Consider: FreeStyle Libre 3 (longest sensor duration, cost-effective)
- Alternative: Dexcom G7 (fastest warm-up, most accurate)
Type 2 Diabetes Not Using Insulin:
- Consider: Dexcom Stelo (OTC, no prescription needed)
- Alternative: FreeStyle Libre 3 (if prescription accessible)
Active Lifestyle/Athletes with Diabetes:
- Consider: Medtronic Guardian 4 (exercise modes, predictive alerts)
- Alternative: Dexcom G7 (real-time sharing, fast warm-up)
Step 2: Assess Cost and Insurance Coverage
Medicare Part B Beneficiaries:
- Verify coverage criteria (insulin use, testing frequency ≥4/day)
- Most prescription CGMs covered with documentation
- Dexcom Stelo (OTC) not typically covered
Commercial Insurance:
- Check formulary for preferred CGM brands
- Obtain prior authorization before ordering
- Appeal denials with clinical documentation
No Insurance/High Deductible:
- Dexcom Stelo: Lowest out-of-pocket cost
- FreeStyle Libre 3: Lower cost than Dexcom G7
- Manufacturer assistance programs may reduce costs
Step 3: Consider Integration Needs
Insulin Pump Users:
- Tandem t:slim X2 requires Dexcom G6 integration
- Medtronic MiniMed 780G requires Guardian 4 integration
- Check compatibility before purchasing
Smartphone-Only Users:
- Dexcom G7: No receiver required
- FreeStyle Libre 3: No receiver required
- Dexcom Stelo: Smartphone required (no receiver option)
Family Glucose Sharing:
- Dexcom G7: Up to 10 followers
- FreeStyle Libre 3: Share data via LibreView
- Guardian 4: Limited sharing (primarily pump-focused)
Step 4: Evaluate Lifestyle Factors
Sensor Change Frequency Preference:
- 14-15 days: FreeStyle Libre 3, Dexcom Stelo
- 10 days: Dexcom G7
- 7 days: Guardian 4
Warm-up Time Sensitivity:
- 30 minutes: Dexcom G7, Dexcom Stelo
- 60 minutes: FreeStyle Libre 3
- 2 hours: Guardian 4
Adhesive Sensitivity:
- All CGMs may cause skin reactions
- Trial period recommended before bulk purchasing
- Discuss alternatives with healthcare provider if reactions occur
Your First Week with a CGM: What to Expect
Day 1: Insertion and Activation
- Insertion takes 30-60 seconds (watch manufacturer video first—it helps!)
- Sensor needs 30-60 minute warm-up period before first reading
- Common concern: “It feels weird”—this is normal and disappears within 24 hours as you forget it’s there
Days 2-3: Learning the App
- Spend 10 minutes exploring the app features (don’t try to master everything at once)
- Set your target range with your doctor’s guidance (typically 70-180 mg/dL)
- Tip: Don’t panic over every high or low reading yet—you’re still learning your patterns
Days 4-7: Understanding Your Patterns
- Notice which foods cause spikes (everyone is different—you might be surprised)
- Observe your overnight patterns (this is data you’ve never had before)
- Take screenshots of interesting patterns to discuss with your doctor
Week 2+: Making Adjustments
- Small changes based on data (timing of meals, exercise, medication)
- Share reports with your healthcare team
- Join online communities (r/diabetes, Beyond Type 1) to compare experiences
When to Call Support vs. Troubleshoot Yourself:
Call manufacturer support if:
- Sensor fails within first 24 hours
- Readings are 50+ mg/dL off from finger stick (after warm-up period)
- Adhesive causes severe skin reaction
- App won’t connect to sensor after multiple restart attempts
Troubleshoot yourself first if:
- Readings seem slightly off (wait 24 hours—sensors stabilize after initial day)
- Adhesive edges lifting (use overlay patches like Skin Grip)
- Forgot to scan/check app (set phone reminders until it becomes habit)
Normal learning curve: Most users feel comfortable after 2-3 sensor changes (3-6 weeks).
References
Device Accuracy and Clinical Trials:
- FDA 510(k) Summaries: Dexcom G7 (K213369), FreeStyle Libre 3 (K221503), Guardian 4 (K210691)
- Wadwa RP, et al. Accuracy of a Factory-Calibrated, Real-Time Continuous Glucose Monitoring System During 10 Days of Use in Youth and Adults with Diabetes. Diabetes Technol Ther. 2023.
- Brown SA, et al. Six-Month Randomized, Multicenter Trial of Closed-Loop Control in Type 1 Diabetes. N Engl J Med. 2019;381:1707-1717.
- Forlenza GP, et al. Predictive Low-Glucose Suspend Reduces Hypoglycemia in Adults, Adolescents, and Children with Type 1 Diabetes in an At-Home Randomized Crossover Study. Diabetes Care. 2018.
Type 2 Diabetes CGM Evidence:
- Bolinder J, et al. Novel Glucose-Sensing Technology and Hypoglycaemia in Type 1 Diabetes: A Multicentre, Non-masked, Randomised Controlled Trial. Lancet. 2016;388:2254-2263.
- Haak T, et al. Flash Glucose-Sensing Technology as a Replacement for Blood Glucose Monitoring for the Management of Insulin-Treated Type 2 Diabetes: The REPLACE Trial. Diabetes Ther. 2017;8:55-73.
Medicare Coverage and Guidelines:
- Centers for Medicare & Medicaid Services. Local Coverage Determination (LCD): Glucose Monitors (L33822). Revised 2023.
- CMS Manual System Pub 100-03, Medicare National Coverage Determinations, Section 40.2: Blood Glucose Testing.
Professional Guidelines:
- American Diabetes Association. Standards of Medical Care in Diabetes—2024. Diabetes Care. 2024;47(Suppl 1).
- Peters AL, et al. Diabetes Technology—Continuous Subcutaneous Insulin Infusion Therapy and Continuous Glucose Monitoring in Adults: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2016.
Accuracy Standards:
U.S. Food and Drug Administration. Self-Monitoring Blood Glucose Test Systems for Over-the-Counter Use: Guidance for Industry and FDA Staff. 2020.
ISO 15197:2013. In Vitro Diagnostic Test Systems — Requirements for Blood-Glucose Monitoring Systems for Self-Testing in Managing Diabetes Mellitus.
Last Updated: 10th April ,2026
Medical Reviewer: Dr. Rishav Das, M.B.B.S.





