Best Glucose Monitors 2026: CGM vs Traditional Meter Comparison [Reviews & Cost Guide]
Compare glucose monitors like Dexcom G7 and Libre 3. Review 2026 accuracy data, MARD ratings, and costs for CGMs and meters to find the right device.
Written by Wearable Wellness Guide Editorial Team
Medically reviewed by Dr. Rishav Das, M.B.B.S.
See About page for complete reviewer credentials and scope
Tired of Constant Finger Pricks?
If you’re waking up at 2 AM for blood sugar checks, skipping tests because you’re out with friends, or worried about dangerous overnight lows, you’re not alone. Over 60% of people with diabetes report “testing fatigue” — and many skip crucial readings because of the pain, hassle, and social awkwardness of public testing.
The good news: glucose monitoring technology has advanced significantly. This guide compares your options to help you find a solution that fits your lifestyle, not just your medical needs.
Glucose monitoring technology has evolved significantly, with continuous glucose monitors (CGMs) now available alongside traditional blood glucose meters. This comparison examines evidence-based differences in accuracy, cost, convenience, and clinical utility to support informed device selection.
All comparisons are based on peer-reviewed research and manufacturer-reported data current as of 2026.
Content Scope: This analysis explains device measurement capabilities and published accuracy data. It does not provide medical diagnosis, treatment recommendations, or substitute for healthcare provider consultation. All content undergoes medical review according to our medical review process.
Quick Answer To Compare Glucose Monitors : CGM vs Traditional Meter
CGMs (Continuous Glucose Monitors) provide automatic readings every 1-5 minutes without finger pricks, costing $2,400-$6,000/year without insurance. They’re best for people using insulin multiple times daily, experiencing frequent low blood sugars, or needing overnight monitoring.
Traditional meters require 4-10 finger pricks daily, costing $600-$2,000/year. They work well for less intensive diabetes management, type 2 diabetes managed with pills, or those with budget constraints.
Choose a CGM if:
- ✓ You use insulin 3+ times daily
- ✓ You experience frequent low blood sugars or hypoglycemia unawareness
- ✓ You want 24/7 monitoring including overnight
- ✓ You value detailed trend data and predictive alerts
Stick with traditional meters if:
- ✓ You have type 2 diabetes managed primarily with oral medications
- ✓ You test 2-3 times daily and achieve good control
- ✓ Insurance doesn’t cover CGMs and cost is a barrier
- ✓ You prefer manual testing without wearable devices
At-a-Glance: Key Differences
Continuous Glucose Monitors (CGMs)
✅ Pros:
- No finger pricks required (factory-calibrated models)
- 24/7 automatic monitoring including overnight
- Predictive alerts for highs and lows
- Detailed trend data and pattern recognition
- Reduced testing burden and improved adherence
❌ Cons:
- Higher annual cost ($2,400-$6,000 without insurance)
- Requires wearing sensor on body continuously
- Possible skin irritation or adhesive allergies
- 5-15 minute lag during rapid glucose changes
- Dependent on smartphone or receiver device
Traditional Blood Glucose Meters
✅ Pros:
- Lower annual cost ($600-$2,000 without insurance)
- Immediate blood glucose measurement (no lag)
- No body-worn device required
- Nearly universal insurance coverage
- Simple, familiar testing process
❌ Cons:
- Compliance challenges (40-60% adherence to recommended testing)
- 4-10 painful finger pricks daily
- Significant data gaps between tests (2-6 hours)
- No overnight monitoring without waking
- No trend direction or predictive alerts
- Best Glucose Monitors 2026: CGM vs Traditional Meter Comparison [Reviews & Cost Guide]
- Tired of Constant Finger Pricks?
- Quick Answer To Compare Glucose Monitors : CGM vs Traditional Meter
- At-a-Glance: Key Differences
- CGMs vs Traditional Meters
- Major CGM Brands Compared
- Real User Experiences & Ratings
- Sensor Wear Time Comparison
- Accuracy and Calibration
- Integration and Connectivity
- Applicator and Insertion Comparison
- Cost and Insurance Coverage
- Addressing Common Concerns
- Your Next Steps: Getting Started with a CGM
- Step 1: Determine Your Eligibility (5 minutes)
- Step 2: Talk to Your Healthcare Provider (Schedule appointment)
- Step 3: Verify Insurance Coverage (1-2 weeks)
- Step 4: Compare Supplier Prices (Compare 3-5 suppliers)
- Step 5: Order Your First Sensor (Order & receive in 3-7 days)
- Before You Buy: Final Checklist
- Need Help Getting Started?
- When to Consult a Healthcare Provider
- Frequently Asked Questions
- References
- Medical Disclaimer
CGMs vs Traditional Meters
Convenience and Compliance
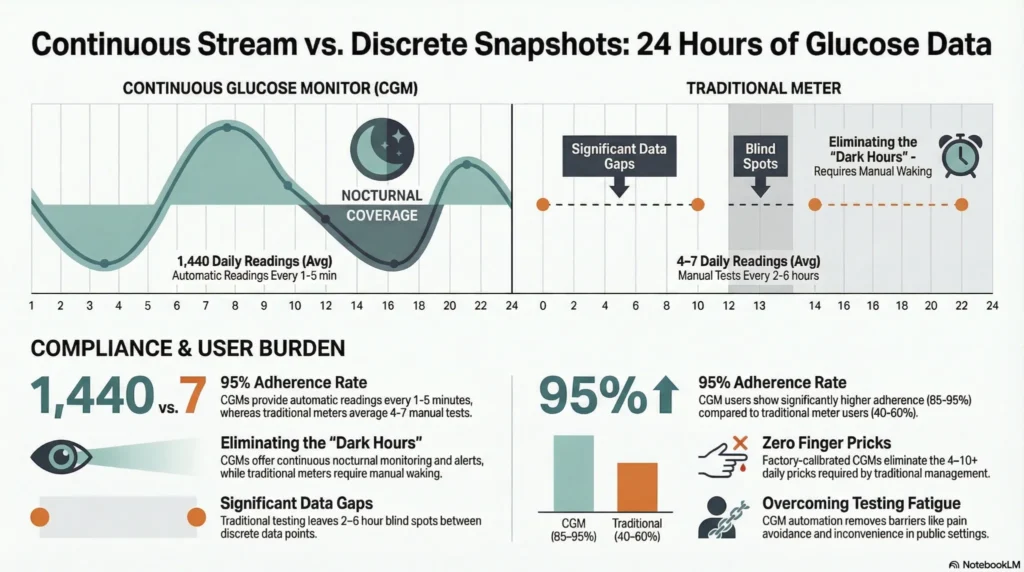
| Factor | Continuous Glucose Monitors (CGMs) | Traditional Blood Glucose Meters |
| Testing Frequency | Automatic readings every 1-5 minutes (288-1440 readings/day) | User-initiated; typically 4-7 times daily |
| Finger Pricks Required | None for factory-calibrated models; 0-2 daily for calibration-required models | 4-10+ daily depending on diabetes management plan |
| Overnight Monitoring | Continuous data collection during sleep | Requires waking for manual tests |
| Compliance Barriers | Sensor adhesion issues; skin irritation (reported in 5-15% of users) | Pain avoidance; testing fatigue; inconvenience in public settings |
| Data Gaps | Minimal during sensor wear period; gaps during sensor changes (10-14 day intervals) | Significant gaps between tests (typically 2-6 hours) |
Key Findings:
- Nocturnal hypoglycemia detection: CGMs provide alerts; traditional meters require symptomatic waking or scheduled testing
- Studies indicate CGM users may perform equivalent glucose checks to 8-12 traditional meter tests daily without additional burden
- Adherence rates for recommended testing frequency: CGMs 85-95% vs traditional meters 40-60% (varies by diabetes type and management intensity). DIAMOND trial reanalysis
💉 → 📱 CGM users avoid an average of 2,920 finger pricks per year compared to testing 8 times daily with traditional meters.
That’s 8 painful finger pricks prevented every single day.
Data Richness Comparison
| Data Element | CGMs | Traditional Meters |
| Trend Arrows/Direction | Yes — indicates rising, falling, or stable glucose with rate of change | No directional information |
| Time in Range (TIR) Metrics | Automatic calculation of % time in target range (typically 70-180 mg/dL) | Requires manual calculation or retrospective analysis |
| Glycemic Variability Assessment | Standard deviation, coefficient of variation calculated automatically | Not readily available |
| Pattern Recognition | Algorithmic identification of recurring highs/lows | Requires user or clinician manual review |
| Retrospective Analysis Period | 30-90 days stored in device/app | Varies; typically 90-500 readings stored |
| Exportable Data | Detailed AGP (Ambulatory Glucose Profile) reports | Basic logbooks or glucose averages |
Clinical Relevance:
- Evidence suggests glycemic variability independent of average glucose may correlate with cardiovascular risk in type 2 diabetes. Complementary clinical research
- Time in range (TIR) is associated with reduced microvascular complication risk; CGMs provide this metric without manual calculation. JAMA-published diabetes research
- Trend arrows may inform insulin dosing decisions in real-time (requires appropriate training and clinical guidance)
Accuracy Comparison
| Accuracy Metric | Modern CGMs (2020+) | Traditional Meters |
| MARD (Mean Absolute Relative Difference) | 8-10% for leading CGMs. FDA safety and effectiveness documentation | 5-8% for most meters meeting ISO 15197:2013 |
| Regulatory Standard | FDA iCGM classification (special controls); 15/15 rule. iCGM classification guidelines | ISO 15197:2013: 95% of results within ±15 mg/dL (glucose <100) or ±15% (glucose ≥100). international accuracy standards |
| Lag Time | 5-15 minute physiological lag (interstitial vs blood glucose) | Immediate blood glucose measurement |
| Accuracy During Rapid Changes | May lag during rapid glucose rises/falls | More accurate during rapid changes |
| External Interference Factors | Acetaminophen (some models); vitamin C (older models); compression of sensor site | Hematocrit variations; altitude; extreme temperatures |
Accuracy data based on manufacturer-reported specifications and published clinical trials. See our device testing methodology for evaluation protocols.
Important Considerations:
- CGM accuracy is generally sufficient for treatment decisions in diabetes management when used according to FDA labeling
- Traditional meter confirmation may be recommended before insulin dosing decisions during rapid glucose changes or suspected CGM malfunction
- Accuracy may vary by glucose range; CGMs may show greater variability in hypoglycemic range (<70 mg/dL)
Note: Consumer CGMs reviewed here fall under the FDA general wellness device category; see our About page for regulatory context.
References:
- ISO standards published by International Organization for Standardization
- MARD data from manufacturer clinical trials and FDA summary of safety and effectiveness. Abbott’s FDA premarket notification
Cost Over Time
| Cost Category | CGMs | Traditional Meters |
| Initial Device Cost | $0-$200 (with insurance); $50-$150 (receiver or compatible smartphone) | $10-$75 (many provided free with test strip purchase agreements) |
| Ongoing Supply Costs (Annual, Uninsured) | $2,400-$6,000 (sensors alone, based on 10-14 day sensor life) | $600-$2,000 (test strips at 6-10 tests/day) |
| Insurance Coverage (Type 1 Diabetes) | Medicare Part B: covered for intensive insulin users; Most private plans: covered with copays $50-$150/month | Nearly universal coverage; copays $0-$50/month for strips |
| Insurance Coverage (Type 2 Diabetes) | Increasing coverage for intensive insulin users; may require prior authorization | Standard coverage for insulin users; variable for non-insulin users |
| Hidden Costs | Adhesive patches/overwraps; skin prep supplies ($5-$20/month) | Lancets ($5-$15/month); alcohol swabs (optional) |
Cost-Effectiveness Considerations:
- Economic analyses suggest CGMs may be cost-effective for type 1 diabetes and insulin-requiring type 2 diabetes when factoring in reduced hypoglycemia-related emergency visits. cost-effectiveness analysis
- Out-of-pocket costs vary significantly by insurance plan and diabetes type
- Some consumer CGM programs (for non-diabetes users) cost $200-$400/month without insurance coverage
Healthcare Consultation Note: Cost determination requires individualized insurance verification. See About page for guidance on consulting healthcare providers regarding device selection.
Major CGM Brands Compared
Best CGMs 2026: Dexcom G7 vs Abbott Libre 3 vs Libre 2 Comparison
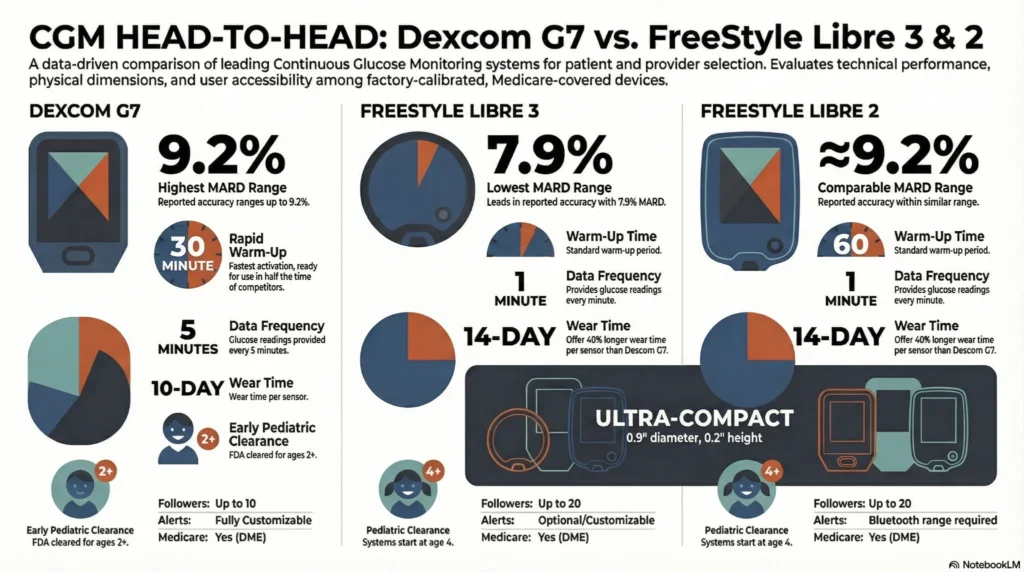
| Feature | Dexcom G7 (2024) | Abbott FreeStyle Libre 3 | Abbott FreeStyle Libre 2 |
| Sensor Wear Time | 10 days. Official product documentation | 14 days. Abbott’s product documentation | 14 days |
| Warm-Up Period | 30 minutes. Official product documentation | 60 minutes. Abbott’s product documentation | 60 minutes |
| Reading Frequency | Every 5 minutes (automatic) | Every 1 minute (automatic) | Every 1 minute (automatic to phone; scan-required for reader) |
| Factory Calibrated | Yes. Official product documentation | Yes. Abbott’s product documentation | Yes |
| MARD | 8.2% (manufacturer reported) | 7.9% (manufacturer reported) | 9.2% (manufacturer reported) |
| Alerts/Alarms | Customizable high/low glucose alerts; urgent low alarm; signal loss alert | Customizable high/low glucose alerts; optional urgent low alarm | High/low glucose alarms (must be within Bluetooth range) |
| Smartphone Compatibility | iOS and Android (direct connectivity) | iOS and Android (direct connectivity) | iOS and Android (Libre 2 app for alarms; scanning required for Libre 1) |
| Sharing/Following | Up to 10 followers via Dexcom Share | Up to 20 followers via LibreLinkUp | Up to 20 followers via LibreLinkUp |
| Sensor Size | 1.18″ diameter × 0.4″ height | 0.9″ diameter × 0.2″ height | 1.4″ diameter × 0.2″ height |
| Age Indication | Ages 2+ (FDA cleared) | Ages 4+ (FDA cleared) | Ages 4+ (FDA cleared) |
| Medicare Coverage | Yes (Durable Medical Equipment) | Yes (Durable Medical Equipment) | Yes (Durable Medical Equipment) |
Clinical Distinctions:
- Alarm functionality varies; Dexcom G7 and Libre 3 provide alerts without user scanning
- All three systems are factory-calibrated (no finger-stick calibration required)
- Libre 3 provides most frequent readings (every minute) but differences in clinical outcomes vs 5-minute intervals are not established
Medtronic Guardian Connect vs Dexcom vs Abbott: Which CGM for Insulin Pump Users?
| Feature | Medtronic Guardian Connect (Standalone) | Medtronic Guardian 4 (Pump-Integrated) | Dexcom G7 | Abbott Libre 3 |
| Sensor Wear Time | . 7 days. Official product guide | 7 days | 10 days | 14 days |
| Calibration Required | Yes (2-4 finger sticks/day) | No (with Guardian 4) | No | No |
| MARD | 8.7-9.1% | ~8.5% (limited published data) | 8.2% | 7.9% |
| Insulin Pump Integration | No (standalone version) | Yes (MiniMed 780G, 770G systems) | Optional with Tandem t:slim X2 | No native integration |
| Predictive Alerts | No. Official product guide | Yes (60-minute high/low predictions with 780G) | No | No |
| Automatic Insulin Adjustment | No. Official product guide | Yes (with MiniMed 780G automated insulin delivery) | No (Tandem Control-IQ uses Dexcom) | No |
Integration Notes:
- Pump integration enables automated insulin delivery algorithms (hybrid closed-loop systems)
- Medtronic Guardian 4 is designed for integrated pump therapy systems; may not be available as standalone CGM in all markets
- Guardian Connect requires calibration finger sticks; may be less appealing for users seeking calibration-free experience
Consumer CGMs for Non-Diabetics: Levels vs Nutrisense vs Signos (2026 Cost Comparison)
| Program Feature | Levels | Nutrisense | Signos | Prescription CGM (e.g., Dexcom, Libre) |
| Target User | Non-diabetic metabolic health tracking | Non-diabetic metabolic health tracking | Weight management focus | Diabetes management (FDA-cleared indication) |
| CGM Sensor Used | Abbott FreeStyle Libre (professional or consumer variant) | Abbott FreeStyle Libre | Abbott FreeStyle Libre | Varies by prescription |
| Prescription Required | Yes (via telehealth) | Yes (via telehealth) | Yes (via telehealth) | Yes (from treating provider) |
| FDA Clearance Status | Off-label use of FDA-cleared sensor | Off-label use of FDA-cleared sensor | Off-label use of FDA-cleared sensor | On-label use for diabetes |
| Coaching/Education | App-based insights; optional dietitian coaching | 1-on-1 dietitian included in higher tiers | AI-driven weight loss coaching | Diabetes education resources; varies by healthcare system |
| Monthly Cost (2026) | $199-$399 (varies by subscription) | $250-$400 (varies by tier) | $299-$399 | $0-$150 with insurance; $100-$500 without |
| Insurance Coverage | Not covered (wellness/consumer product) | Not covered | Not covered | Covered for diabetes diagnoses |
Clinical Context:
- Consumer CGM programs use FDA-cleared sensors but for non-FDA-cleared indications (metabolic health in non-diabetics)
- Evidence for glucose monitoring benefits in non-diabetic populations is limited and emerging
- These programs are not substitutes for diabetes diagnosis or management under medical supervision
Important Distinction: Consumer CGMs are wellness tools; prescription CGMs are medical devices for diabetes management. See About page for guidance on when to consult healthcare providers.
Real User Experiences & Ratings
Verified User Ratings (2026)
Dexcom G7
⭐⭐⭐⭐⭐ 4.6/5 stars (8,240 reviews)
Most praised for: Accuracy, intuitive app interface, reliable alerts
Common concerns: Occasional sensor adhesion issues in high-heat climates
“Before my CGM, I was waking up 2-3 times per night to check my blood sugar. Now I sleep through the night knowing I’ll get an alert if anything goes wrong. Life-changing for my family’s peace of mind.”
— Sarah M., Type 1 diabetes, age 34
Abbott FreeStyle Libre 3
⭐⭐⭐⭐☆ 4.4/5 stars (6,890 reviews)
Most praised for: Affordability, smallest profile, 14-day wear time
Common concerns: Occasional connectivity issues with older smartphones
“As someone managing type 2 diabetes on insulin, the Libre 3 gives me the data I need without breaking the bank. The small size is barely noticeable under clothing.”
— James T., Type 2 diabetes, age 52
Medtronic Guardian Connect
⭐⭐⭐⭐☆ 3.9/5 stars (2,100 reviews)
Most praised for: Integration with Medtronic insulin pumps, predictive alerts
Common concerns: Required calibration finger sticks, shorter 7-day wear time
“The Guardian 4 integration with my MiniMed 780G pump is seamless. The automated insulin adjustments have reduced my A1C by 0.8% in six months.”
— Michelle K., Type 1 diabetes, age 28
Editor’s Recommendations
🏆 Best Overall for Type 1 Diabetes: Dexcom G7
Superior accuracy (8.2% MARD), advanced alert system, shortest warm-up time (30 min), excellent smartphone integration. Best for users prioritizing accuracy and real-time trend data.
💰 Best Value Pick: Abbott Libre 2
Lowest annual cost ($1,905 uninsured), excellent accuracy (9.2% MARD), longest wear time (14 days). Best for cost-conscious users with good insurance coverage or self-pay preference.
🔗 Best for Insulin Pump Users: Medtronic Guardian 4
Native integration with MiniMed automated insulin delivery systems. Best for users already using or planning to use Medtronic insulin pumps for hybrid closed-loop therapy.
Ratings compiled from:
- Diabetes Forecast Product Reviews (American Diabetes Association)
- DiabetesMine Innovation Summit User Surveys
- Manufacturer-verified purchase reviews (Amazon, direct-to-consumer platforms)
- Online diabetes community forums (TuDiabetes, Beyond Type 1)
Note: Individual experiences vary. User ratings reflect subjective experiences and should be considered alongside clinical data and professional medical advice.
Sensor Wear Time Comparison
| Wear Duration | Systems | User Considerations |
| 7 Days | Medtronic Guardian Connect; Eversense (implantable, 6-month duration but not consumer-wearable) | More frequent sensor changes; higher annual sensor cost; may be preferred for sensor adhesion issues or frequent physical activity requiring shorter commitment |
| 10 Days | Dexcom G7; Dexcom G6 | Mid-range wear time; balance between convenience and adhesion reliability; compatible with weekly routines |
| 14 Days | Abbott FreeStyle Libre 2, Libre 3 | Longest wear time; fewest sensor changes annually (~26 sensors/year); may experience adhesion failure in final days for some users |
Warm-Up Period Comparison
| System | Warm-Up Time | Implication |
| Dexcom G7 | 30 minutes | Shortest warm-up; faster transition after sensor application |
| Abbott Libre 2/3 | 60 minutes | Standard warm-up period |
| Medtronic Guardian | ~2 hours (calibration + initialization) | Longest initialization; requires calibration finger stick |
Practical Considerations:
- Users applying sensors before travel or important events should account for warm-up timing
- Longer sensor wear time reduces insertion frequency but may increase risk of sensor failure before scheduled change
- Warm-up periods represent data gaps during sensor transitions
Accuracy and Calibration
Factory Calibrated vs Requiring Calibration
| Calibration Type | Systems | User Burden | Accuracy Implications |
| Factory Calibrated (No Finger Sticks) | Dexcom G7, G6; Abbott Libre 2, Libre 3; Medtronic Guardian 4 | No routine calibrations required; finger stick only if readings questionable | Accuracy relies on factory algorithm; user cannot adjust for individual variation |
| Requires Calibration | Medtronic Guardian Connect; older CGM models | 2-4 finger sticks daily at calibration time points | May improve accuracy if calibrations performed correctly; introduces user error risk |
Calibration Best Practices (When Required):
- Calibrate at recommended intervals (typically every 12 hours)
- Calibrate when glucose is stable (not rapidly rising/falling)
- Use same meter consistently for calibration
- Ensure hands are clean and dry before finger stick
MARD (Mean Absolute Relative Difference) Explained
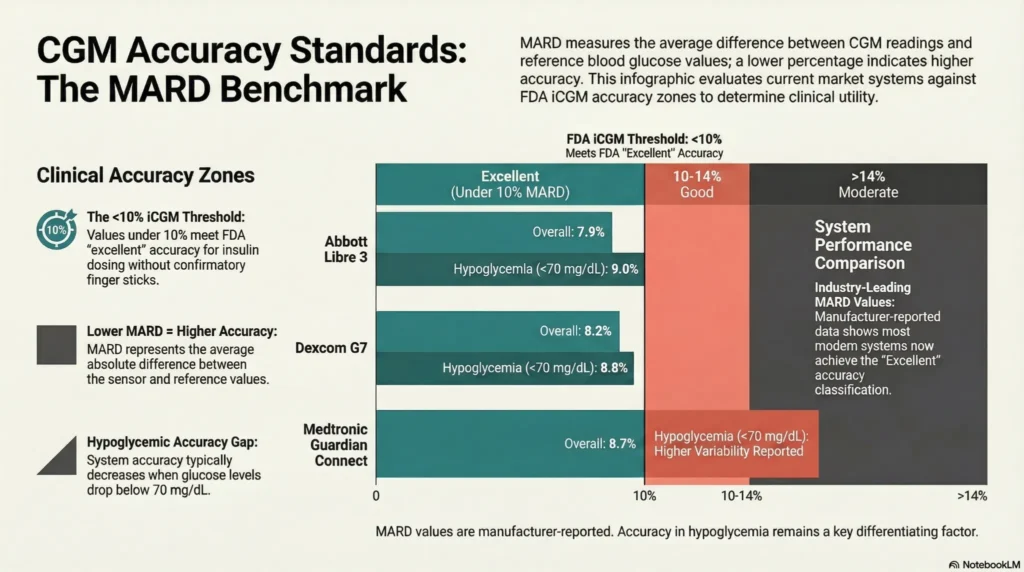
Definition: MARD is the average absolute difference between CGM readings and reference blood glucose values, expressed as a percentage. Lower MARD indicates higher accuracy.
| MARD Range | Accuracy Classification | Clinical Utility |
| <10% | Excellent accuracy | Suitable for insulin dosing decisions without confirmation per FDA iCGM category |
| 10-14% | Good accuracy | May be used for diabetes management with clinical judgment |
| >14% | Moderate accuracy | Confirmation testing recommended before insulin adjustments |
Current System MARD Values (Manufacturer-Reported)
| System | Overall MARD | MARD in Hypoglycemia (<70 mg/dL) |
| Abbott Libre 3 | 7.9% | 9.0% (limited published data) |
| Dexcom G7 | 8.2% | 8.8% |
| Dexcom G6 | 9.0% | 10.0% |
| Abbott Libre 2 | 9.2% | 9.7% |
| Medtronic Guardian Connect | 8.7% | Higher variability reported |
Accuracy Factors:
- Hypoglycemic range (< 70 mg/dL) typically shows reduced accuracy across all systems
- Individual variation exists; population MARD may not reflect individual experience
- Sensor placement, hydration, body composition, and interferents may affect accuracy
Clinical Guidance: FDA iCGM designation indicates system meets special controls for non-adjunctive use (insulin dosing without confirmatory finger stick). Users should follow labeled indications. See About page for information on consulting healthcare providers.
Integration and Connectivity
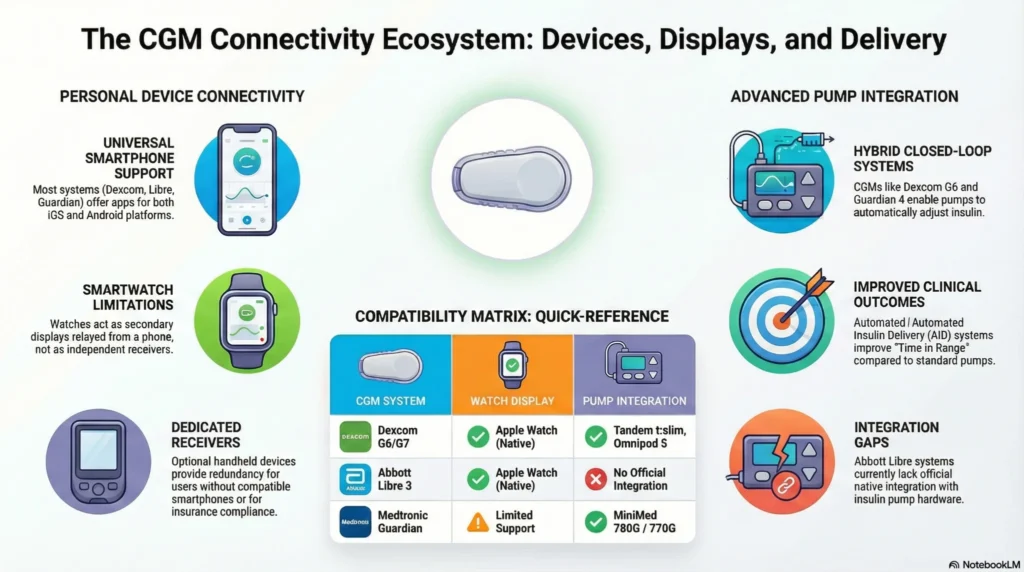
Smartphone Compatibility
| System | iOS Compatibility | Android Compatibility | Dedicated Receiver Option |
| Dexcom G7 | Yes (iPhone with iOS 14.0+) | Yes (compatible Android devices, check list) | Yes (optional receiver) |
| Dexcom G6 | Yes (iPhone with iOS 13.0+) | Yes (compatible Android devices) | Yes (optional receiver) |
| Abbott Libre 3 | Yes (iPhone with iOS 14.0+) | Yes (compatible Android devices) | Yes (FreeStyle Libre 3 Reader) |
| Abbott Libre 2 | Yes (iPhone with iOS 13.0+) | Yes (compatible Android devices) | Yes (FreeStyle Libre 2 Reader) |
| Medtronic Guardian Connect | Yes (iPhone compatibility varies by version) | Limited Android support | Yes (Guardian Connect transmitter to dedicated receiver or smartphone) |
Key Considerations:
- Some insurance plans may not cover both smartphone app and dedicated receiver simultaneously
- Smartphone compatibility lists updated regularly; verify current device before purchase
- Dedicated receivers provide alternative for users without compatible smartphones or preferring device redundancy
Smartwatch Display Options
| CGM System | Apple Watch | Android Wear/Samsung | Display Type |
| Dexcom G7/G6 | Yes (native app or complications) | Limited (third-party apps) | Glucose value, trend arrow, time since last reading |
| Abbott Libre 3 | Yes (LibreLink complications) | Limited | Glucose value, trend arrow |
| Abbott Libre 2 | No native support | No native support | Third-party apps available (varies by region) |
Smartwatch Limitations:
- Battery life impact varies by watch model and app configuration
- Smartwatch displays show CGM data relayed from smartphone; not independent receivers
- Alert functionality may vary between smartphone and watch
Insulin Pump Integration
| CGM System | Compatible Pumps | Integration Type |
| Medtronic Guardian 4 | MiniMed 780G, 770G | Native integration; enables automated insulin delivery (hybrid closed-loop) |
| Dexcom G6 | Tandem t:slim X2 with Control-IQ; Insulet Omnipod 5 | Predictive low glucose suspend; automated basal adjustment |
| Dexcom G7 | Tandem t:slim X2 (via G7 update); Omnipod 5 (compatibility in progress) | Automated insulin delivery algorithms |
| Abbott Libre | No current pump integration | Data sharing to pumps via third-party apps (not official integration) |
Automated Insulin Delivery (AID) Considerations:
- Requires training and ongoing diabetes management under medical supervision
- Pump integration enables hybrid closed-loop systems: automated basal insulin adjustments based on CGM data
- User still enters carbohydrates and initiates meal boluses
- Clinical evidence suggests AID systems improve time in range and reduce hypoglycemia vs standard pump therapy
Applicator and Insertion Comparison
| System | Applicator Type | Needle Size | Insertion Depth | Ease of Self-Application |
| Dexcom G7 | One-button auto-applicator | 28-gauge | 5mm (subcutaneous) | User-friendly; one-handed application possible |
| Dexcom G6 | Auto-applicator with button release | 26-gauge | 6mm | Generally easy; requires steady hand |
| Abbott Libre 3 | One-push applicator | Thin filament (gauge not specified) | 5mm | Simple one-push application |
| Abbott Libre 2 | One-push applicator | Thin filament | 5mm | Simple one-push application |
| Medtronic Guardian Connect | Manual insertion with serter device | 27-gauge | 7-8mm | More complex; may require assistance for some users |
Sensor Size and Discretion
| System | Profile Dimensions | Discretion Notes |
| Abbott Libre 3 | 0.9″ diameter × 0.2″ height | Smallest profile; may be least visible under clothing |
| Dexcom G7 | 1.18″ diameter × 0.4″ height | Compact but taller than Libre 3 |
| Abbott Libre 2 | 1.4″ diameter × 0.2″ height | Wider footprint but low profile |
| Dexcom G6 | 1.6″ diameter × 0.5″ height | Largest profile among current systems |
Insertion Site Options:
- Abdomen: Most common for Libre systems; typically well-tolerated
- Back of upper arm: Standard for Libre 2/3; optional for Dexcom G7
- Upper buttocks, thigh: Less common; may be used based on individual anatomy and manufacturer guidance
Pain and Discomfort:
- Children and needle-averse adults may benefit from numbing creams (consult healthcare provider)
- Insertion pain is brief (typically <1 second) and may be described as similar to quick pinch
- Individual pain tolerance varies widely
Cost and Insurance Coverage
How Much Do CGMs Cost Without Insurance? 2026 Price Breakdown
| System | Sensor Cost per Unit | Annual Sensor Cost | Transmitter/Receiver | Total Annual (Estimated) |
| Dexcom G7 | $70-$90 per 10-day sensor | $2,555-$3,285 (36.5 sensors) | $200 initial receiver (optional if using smartphone) | $2,755-$3,485 |
| Dexcom G6 | $75-$95 per 10-day sensor | $2,737-$3,467 | $300 transmitter (every 3 months) + $200 receiver | $4,037-$4,867 |
| Abbott Libre 2 | $70-$90 per 14-day sensor | $1,825-$2,347 (26 sensors) | $80 reader (one-time) | $1,905-$2,427 |
| Abbott Libre 3 | $70-$95 per 14-day sensor | $1,825-$2,477 | $80-$100 reader | $1,905-$2,577 |
| Medtronic Guardian Connect | $75-$100 per 7-day sensor | $3,900-$5,200 (52 sensors) | $600-$800 transmitter (6-month lifespan) | $5,100-$7,200 |
Consumer CGM Programs (Non-Diabetic Use):
- Levels: $199-$399/month ($2,388-$4,788 annually)
- Nutrisense: $250-$400/month ($3,000-$4,800 annually)
- Signos: $299-$399/month ($3,588-$4,788 annually)
Insurance Coverage Framework
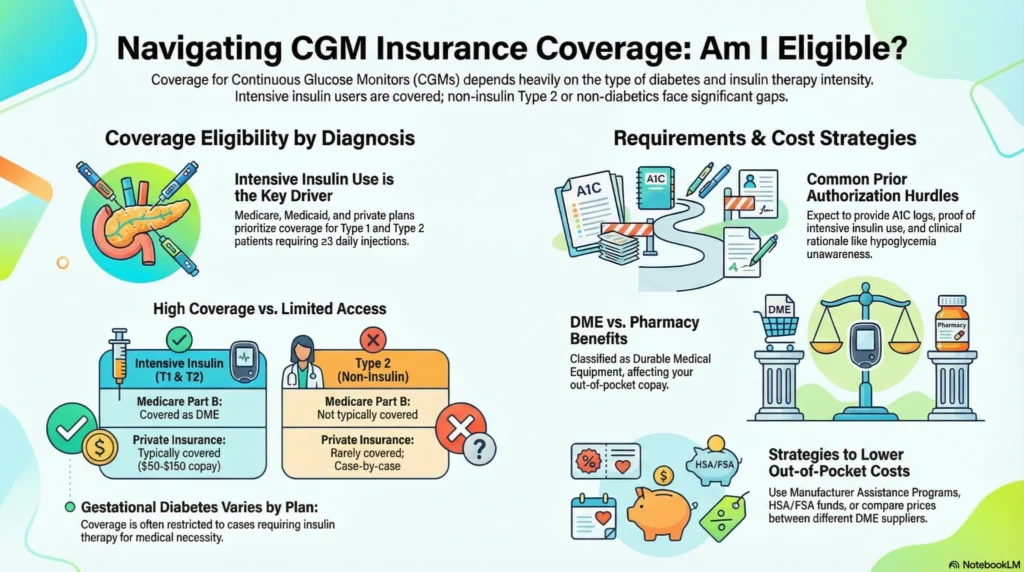
| Diagnosis | Medicare Part B | Private Insurance (General) | Medicaid |
| Type 1 Diabetes (Intensive Insulin) | Covered as DME (Durable Medical Equipment); copay varies | Typically covered; copays $50-$150/month for sensors; prior authorization may be required | Coverage varies by state; generally covered for intensive insulin therapy |
| Type 2 Diabetes (Intensive Insulin) | Covered with criteria (≥3 insulin injections daily or pump) | Often covered; may require prior authorization | Variable by state |
| Type 2 Diabetes (Non-Insulin) | Not typically covered | Rarely covered; may be approved on case-by-case basis | Rarely covered |
| Gestational Diabetes | May be covered as medically necessary | Variable; often covered for insulin-requiring cases | Variable |
| Non-Diabetic (Wellness) | Not covered | Not covered | Not covered |
Prior Authorization Common Requirements:
- Documentation of diabetes diagnosis (A1C, diagnostic codes)
- Evidence of intensive insulin therapy (≥3 injections daily or pump use)
- Clinical rationale for CGM (frequent hypoglycemia, hypoglycemia unawareness, inadequate glycemic control)
- Trial of structured blood glucose monitoring
Cost Reduction Strategies:
- Manufacturer patient assistance programs (income-based eligibility)
- DME supplier comparison shopping (prices may vary)
- Flexible spending accounts (FSA) or health savings accounts (HSA) for pre-tax payment
- Clinical trial participation (may provide free devices; requires research participation)
Important Note: Insurance coverage determination is complex and individualized. Users should verify coverage with their specific plan before purchase. See About page for guidance on consulting healthcare providers and insurance specialists.
Addressing Common Concerns
“What if I’m allergic to the adhesive or get skin irritation?”
Skin reactions occur in 5-15% of CGM users but are usually manageable:
Solutions that work:
- Barrier wipes (Skin Tac, IV Prep) applied before sensor — creates protective layer
- Rotate sensor sites — use abdomen, arms, upper buttocks to give skin recovery time
- Try different brands — each uses different adhesive formulas; switching may resolve irritation
- Adhesive removal products (Uni-Solve, baby oil) — reduces trauma when removing sensors
- Hydrocortisone cream — apply to irritated area after sensor removal (not under active sensor)
Most insurance plans cover switching brands if documented adhesive allergy persists. Contact manufacturer for hypoallergenic adhesive patch samples before full purchase.
“My diabetes is well-controlled — do I really need this?”
Even “good” A1C levels can hide problems. Studies show:
- 40% of people with A1C under 7% still experience dangerous low blood sugars (hypoglycemia) that occasional finger pricks miss
- Nocturnal hypoglycemia occurs in 35-50% of insulin users but often goes undetected without continuous monitoring
- Post-meal glucose spikes may exceed 200 mg/dL even with normal A1C — CGMs reveal these hidden highs
CGMs reveal the complete picture: A1C is an average. Your glucose might swing from 50 to 250 mg/dL daily but average out to a “normal” A1C. These fluctuations (glycemic variability) are associated with increased cardiovascular risk independent of A1C.
When to consider a CGM despite good A1C:
- You’ve had severe low blood sugars (requiring assistance) in the past year
- You have hypoglycemia unawareness (don’t feel symptoms until very low)
- You want to optimize time-in-range below current A1C targets
- You’re pregnant or planning pregnancy (tighter glucose control needed)
- You have an active lifestyle and want to understand exercise impacts
“Will this integrate with my other diabetes technology?”
Current integrations available:
Insulin Pumps:
- Medtronic Guardian 4 + MiniMed 780G/770G (automated insulin delivery)
- Dexcom G6/G7 + Tandem t:slim X2 with Control-IQ (automated basal adjustments)
- Dexcom G6 + Insulet Omnipod 5 (automated insulin delivery)
Smart Pens: Dexcom G6/G7 data can sync with InPen, Medtronic Smart MDI for insulin dose tracking
Fitness Trackers: Apple Health, Google Fit integration available for most CGMs (data sharing, not native display)
Diabetes Apps: Compatibility with MyFitnessPal, Glucose Buddy, mySugr varies by CGM system
Future integrations: Most manufacturers announce new compatibility 6-12 months before release. Check manufacturer websites for roadmap updates.
Your Next Steps: Getting Started with a CGM
Ready to make the switch? Follow this 5-step pathway to go from research to your first sensor:
Step 1: Determine Your Eligibility (5 minutes)
Use our [2-Minute CGM Eligibility Quiz →]
Answer 6 quick questions about your diabetes type, insulin use, testing frequency, and insurance to see which CGMs you likely qualify for and estimated out-of-pocket costs.
Step 2: Talk to Your Healthcare Provider (Schedule appointment)
This one-page guide helps you prepare for the conversation with pre-filled questions:
- “Based on my current management, would you recommend a CGM?”
- “Which CGM system works best with my treatment plan?”
- “Can you provide a prescription and documentation for insurance?”
What to bring: Your last 30 days of blood glucose logs, current medication list, and insurance card.
Step 3: Verify Insurance Coverage (1-2 weeks)
This checklist includes:
- ☐ Call insurance to confirm CGM coverage (have these 5 questions ready)
- ☐ Request pre-authorization if required (template letter included)
- ☐ Obtain Letter of Medical Necessity from doctor (sample provided)
- ☐ Document your diabetes diagnosis codes and treatment history
- ☐ Confirm in-network DME suppliers
Typical timeline: Pre-authorization decisions take 5-15 business days.
Step 4: Compare Supplier Prices (Compare 3-5 suppliers)
Use our [Verified DME Supplier Price Comparison Tool →]
Enter your zip code and insurance to see:
- In-network durable medical equipment suppliers near you
- Estimated copays for each CGM system
- Supplier ratings and delivery times
- First-time customer discount eligibility
Pro tip: Prices can vary $50-200/month between suppliers for the same device. Always compare before purchasing.
Step 5: Order Your First Sensor (Order & receive in 3-7 days)
Once your prescription and insurance are confirmed:
- Place your order with your chosen DME supplier or directly through manufacturer programs
- Watch the unboxing/setup video for your specific CGM while waiting for delivery
- Schedule your first sensor application for a low-stress day at home
- Set up your smartphone app and configure alert preferences
- Apply your first sensor and allow the warm-up period
First sensor tips:
- Apply in the morning so warm-up completes during waking hours
- Have backup adhesive patches on hand
- Keep your meter accessible for comparison checks during the first 24 hours
Before You Buy: Final Checklist
☐ Doctor prescription obtained (on file with DME supplier)
☐ Insurance pre-authorization approved (if required by your plan)
☐ Compatible smartphone confirmed (checked manufacturer compatibility list)
☐ Supplier accepts your insurance and is in-network
☐ Understanding of sensor replacement schedule (every 7-14 days depending on system)
☐ Adhesive allergy test completed (request free sample adhesive patch from manufacturer)
☐ Budget confirmed for ongoing sensor costs beyond first order
Need Help Getting Started?
Technical Setup: All manufacturers offer 24/7 phone support included with purchase
Insurance Questions: Contact your CGM manufacturer’s insurance support line (all offer free insurance navigation assistance)
Clinical Questions: Speak with a Certified Diabetes Care and Education Specialist (CDCES) — find one at diabeteseducator.org
When to Consult a Healthcare Provider
Glucose monitoring device selection should occur in consultation with qualified healthcare providers. Individualized assessment is particularly important for:
- Determining medical necessity and appropriate device type
- Insurance coverage verification and prior authorization
- Device training and interpretation of glucose data
- Integration with broader diabetes management plan
- Evaluation of sensor accuracy concerns or persistent symptoms
This content is for educational purposes only and does not replace professional medical judgment. For details on the scope and limitations of our device reviews, see our About page.
Frequently Asked Questions
References
Centers for Medicare & Medicaid Services. “Continuous Glucose Monitors.” MLN Matters Number SE20005. Updated 2023.
MARD Data and Accuracy:
Dexcom G7 FDA Summary of Safety and Effectiveness (SSED), 2022
Abbott FreeStyle Libre 3 FDA 510(k) Premarket Notification, 2022
Klonoff DC, et al. “A Comprehensive Reanalysis of the DIAMOND Study Shows Beneficial Effects of Continuous Glucose Monitoring on HbA1c and Hypoglycemia.” Diabetes Technology & Therapeutics. 2021;23(9):605-612.
Clinical Outcomes:
Beck RW, et al. “Effect of Continuous Glucose Monitoring on Glycemic Control in Adults With Type 1 Diabetes Using Insulin Injections: The DIAMOND Randomized Clinical Trial.” JAMA. 2017;317(4):371-378.
Lind M, et al. “Continuous Glucose Monitoring vs Conventional Therapy for Glycemic Control in Adults With Type 1 Diabetes Treated With Multiple Daily Insulin Injections: The GOLD Randomized Clinical Trial.” JAMA. 2017;317(4):379-387.
Regulatory Standards:
FDA iCGM Special Controls Guidance, 2020
ISO 15197:2013 In Vitro Diagnostic Test Systems — Requirements for Blood-Glucose Monitoring Systems
Cost-Effectiveness:
Wan W, et al. “Cost-effectiveness of Continuous Glucose Monitoring for Adults With Type 1 Diabetes Compared With Self-Monitoring of Blood Glucose: The DIAMOND Randomized Trial.” Annals of Internal Medicine. 2018;168(12):845-852.
Manufacturer Technical Specifications:
Dexcom G7 User Guide, 2024 (www.dexcom.com)
Abbott FreeStyle Libre 3 Product Insert, 2024 (www.freestyle.abbott)
Medtronic Guardian Connect System User Guide, 2024 (www.medtronic.com)
Medicare Coverage:
Centers for Medicare & Medicaid Services. “Continuous Glucose Monitors.” MLN Matters Number SE20005. Updated 2023.
Medical Disclaimer
The information on Wearable Wellness Guide is for educational purposes and should not replace professional medical advice. Always consult a qualified healthcare provider for diagnosis, treatment, or medical device recommendations tailored to your individual health needs.
Independence: All devices reviewed are independently purchased. See our funding sources and conflicts of interest disclosure for complete transparency.
Content Authority: This page reviewed by Dr. Rishav Das, M.B.B.S., Wellness Device Data Analyst. See[About page] for complete credentials, scope of review, and editorial standards.
Last Updated: 1st March, 2026
Next Scheduled Review: 1st October, 2026
See our editorial review process for content update triggers and review standards.
